Successive climate crises in the deep past drove the early evolution and radiation of reptiles
- PMID: 35984885
- PMCID: PMC9390993
- DOI: 10.1126/sciadv.abq1898
Successive climate crises in the deep past drove the early evolution and radiation of reptiles
Abstract
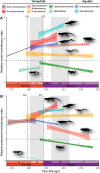
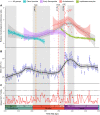
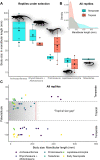
Climate change-induced mass extinctions provide unique opportunities to explore the impacts of global environmental disturbances on organismal evolution. However, their influence on terrestrial ecosystems remains poorly understood. Here, we provide a new time tree for the early evolution of reptiles and their closest relatives to reconstruct how the Permian-Triassic climatic crises shaped their long-term evolutionary trajectory. By combining rates of phenotypic evolution, mode of selection, body size, and global temperature data, we reveal an intimate association between reptile evolutionary dynamics and climate change in the deep past. We show that the origin and phenotypic radiation of reptiles was not solely driven by ecological opportunity following the end-Permian extinction as previously thought but also the result of multiple adaptive responses to climatic shifts spanning 57 million years.
Figures



References
-
- Sinervo B., Méndez-de-la-Cruz F., Miles D. B., Heulin B., Bastiaans E., Villagrán-Santa Cruz M., Lara-Resendiz R., Martínez-Méndez N., Calderón-Espinosa M. L., Meza-Lázaro R. N., Gadsden H., Avila L. J., Morando M., de la Riva I. J., Sepulveda P. V., Rocha C. F. D., Ibargüengoytía N., Puntriano C. A., Massot M., Lepetz V., Oksanen T. A., Chapple D. G., Bauer A. M., Branch W. R., Clobert J., Sites J. W. Jr., Erosion of lizard diversity by climate change and altered thermal niches. Science 328, 894–899 (2010). - PubMed
-
- Meiri S., Bauer A. M., Chirio L., Colli G. R., Das I., Doan T. M., Feldman A., Herrera F. C., Novosolov M., Pafilis P., Pincheira-Donoso D., Powney G., Torres-Carvajal O., Uetz P., van Damme R., Are lizards feeling the heat? A tale of ecology and evolution under two temperatures. Glob. Ecol. Biogeogr. 22, 834–845 (2013).
LinkOut - more resources
Full Text Sources
Miscellaneous

