In Vivo Effects of Conditioned Medium from Human Uterine Cervical Stem Cells in an Ovarian Cancer Xenograft Mouse Model
- PMID: 35985689
- PMCID: PMC9353723
- DOI: 10.21873/cgp.20341
In Vivo Effects of Conditioned Medium from Human Uterine Cervical Stem Cells in an Ovarian Cancer Xenograft Mouse Model
Abstract
Background/aim: Ovarian cancer is the most lethal of all gynecological cancers, despite advances in surgical techniques and medical treatments. During the last years, therapies based on mesenchymal stem cells and particularly their secretome (conditioned medium, CM) have emerged as promising treatments for various types of tumors.
Materials and methods: In the present study, we evaluated the in vivo antitumor effect of human uterine cervical stem cell conditioned medium (hUCESC-CM) after intraperitoneal administration in an ovarian cancer mouse model.
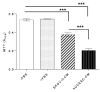
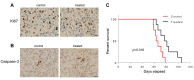
Results: We found that intraperitoneal injection of hUCESC-CM in immunodeficient mice, injected fifty days previously with the human ovarian adenocarcinoma SKOV-3 cell line, significantly reduced abdominal tumor growth, and significantly increased overall survival, compared to control mice.
Conclusion: hUCESC-CM could be an alternative approach to intraperitoneal treatment of ovarian cancer, either administered alone and/or with conventional chemotherapy.
Keywords: Mesenchymal stem cells; conditioned medium; ovarian cancer; secretome.
Copyright© 2022, International Institute of Anticancer Research (Dr. George J. Delinasios), All rights reserved.
Conflict of interest statement
The Authors declare the following competing interests: Francisco J. Vizoso, Roman Perez-Fernandez, and Noemi Eiró are co-inventors of a patent (“Human uterine cervical stem cell population and uses thereof”) owned by GiStem Research, of which some authors are shareholders (JS-L, SS, NE, FV, RP-F, and JS). The founding sponsors had no role in the design of this manuscript, in the collection, analyses, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
Figures

References
-
- Knisely AT, St Clair CM, Hou JY, Collado FK, Hershman DL, Wright JD, Melamed A. Trends in primary treatment and median survival among women with advanced-stage epithelial ovarian cancer in the US from 2004 to 2016. JAMA Netw Open. 2020;3(9):e2017517. doi: 10.1001/jamanetworkopen.2020.17517. - DOI - PMC - PubMed
-
- van Driel WJ, Koole SN, Sikorska K, Schagen van Leeuwen JH, Schreuder HWR, Hermans RHM, de Hingh IHJT, van der Velden J, Arts HJ, Massuger LFAG, Aalbers AGJ, Verwaal VJ, Kieffer JM, Van de Vijver KK, van Tinteren H, Aaronson NK, Sonke GS. Hyperthermic intraperitoneal chemotherapy in ovarian cancer. N Engl J Med. 2018;378(3):230–240. doi: 10.1056/NEJMoa1708618. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
