Translational research in health technologies: A scoping review
- PMID: 35990015
- PMCID: PMC9385029
- DOI: 10.3389/fdgth.2022.957367
Translational research in health technologies: A scoping review
Abstract
Introduction: The current debate on the process of technological innovation points out as a challenge for universities consolidation of competencies that allow the generation and transfer of knowledge to society. The Translational Research (TR) approach has as one of its main objectives the acceleration of the innovation process, based on the transposition from basic science to applied science and innovation, which comprises the different stages of research, development and innovation. The literature points out that the dynamics of translation, which results in new technologies, are complex, transdisciplinary, inter-institutional, systemic, and non-linear. The main objective of this review is to contribute to the adoption of institutional strategies and the formulation of public policies aimed at solving today's social and economic challenges, ensuring access to technologies and sustainability for the health system. The specific objectives were: (i) to systematize studies that characterized translational research in medical devices; (ii) map the challenges for the implementation of translational health research; (iii) contribute to the design of institutional strategies; and (iv) support the formulation of public policies.
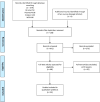
Methods: This study used the scoping review technique, according to PRISMA-ScR and the Joanna Briggs Institute guidelines. Concerning the extraction of relevant articles, the journals indexed in Bireme, Pubmed, Scopus, Web of Science, and Google Scholar were consulted for selecting relevant articles. The search was carried out on November 28, 2021, updated on April 29, 2022, and there were no restrictions as to the year of publication, language or type of analysis. Studies that did not answer the research question were excluded, as they dealt exclusively with the pharmaceutical segment, the translation of knowledge into clinical practice, or addressed the process of translational research applied to specific diseases or technologies.
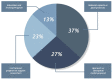
Results: Thirty-three articles were included indicating that the approach of translation of research is multidisciplinary and transdisciplinary and encompasses knowledge and aspects that go beyond basic and applied research and incorporates final steps concerning regulatory aspects, clinical research, market analysis, technology transfer, production and incorporation of technologies into the health system.
Keywords: health innovation; health technologies; medical device; medical research; translational research.
© 2022 Mayrink, Alcoforado, Chioro, Fernandes, Lima, Camargo and Valentim.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Beyond the black stump: rapid reviews of health research issues affecting regional, rural and remote Australia.Med J Aust. 2020 Dec;213 Suppl 11:S3-S32.e1. doi: 10.5694/mja2.50881. Med J Aust. 2020. PMID: 33314144
-
Artificial intelligence technologies and compassion in healthcare: A systematic scoping review.Front Psychol. 2023 Jan 17;13:971044. doi: 10.3389/fpsyg.2022.971044. eCollection 2022. Front Psychol. 2023. PMID: 36733854 Free PMC article.
-
The Effectiveness of Integrated Care Pathways for Adults and Children in Health Care Settings: A Systematic Review.JBI Libr Syst Rev. 2009;7(3):80-129. doi: 10.11124/01938924-200907030-00001. JBI Libr Syst Rev. 2009. PMID: 27820426
-
Towards a European strategy for medicines research (2014-2020): The EUFEPS position paper on Horizon 2020.Eur J Pharm Sci. 2012 Dec 18;47(5):979-87. doi: 10.1016/j.ejps.2012.09.020. Epub 2012 Oct 6. Eur J Pharm Sci. 2012. PMID: 23046836
-
A Framework for Competencies for the Use of Mobile Technologies in Psychiatry and Medicine: Scoping Review.JMIR Mhealth Uhealth. 2020 Feb 21;8(2):e12229. doi: 10.2196/12229. JMIR Mhealth Uhealth. 2020. PMID: 32130153 Free PMC article.
Cited by
-
Enhancing translational medical research through proof-of-concept services: clinicians' perspectives.Sci Rep. 2024 Dec 28;14(1):31108. doi: 10.1038/s41598-024-82401-y. Sci Rep. 2024. PMID: 39730948 Free PMC article.
-
Towards co-design of rehabilitation technologies: a collaborative approach to prioritize usability issues.Front Rehabil Sci. 2024 Feb 21;5:1302179. doi: 10.3389/fresc.2024.1302179. eCollection 2024. Front Rehabil Sci. 2024. PMID: 38450206 Free PMC article.
-
Competency gaps and institutional challenges for translational research in medical devices: insights from Brazilian researchers.BMC Med Educ. 2025 Apr 18;25(1):571. doi: 10.1186/s12909-025-07160-4. BMC Med Educ. 2025. PMID: 40247278 Free PMC article.
References
-
- Freeman C, Technology policy and economic performance. Pinter Pub Ltd; (1987). p.150.
-
- OECD and Eurostat. Oslo manual: guidelines for collecting and interpreting innovation data. In: The measurement of scientific and technological activities. 3rd ed. Paris: OECD Publishing (2005). p. 162. doi:10.1787/9789264013100-en
-
- Freeman C. The ‘National System of Innovation’ in historical perspective. Camb J Econ (1995) 19:5–24. 10.1093/oxfordjournals.cje.a035309 - DOI
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous