Expression of Opsins of the Box Jellyfish Tripedalia cystophora Reveals the First Photopigment in Cnidarian Ocelli and Supports the Presence of Photoisomerases
- PMID: 35991966
- PMCID: PMC9389615
- DOI: 10.3389/fnana.2022.916510
Expression of Opsins of the Box Jellyfish Tripedalia cystophora Reveals the First Photopigment in Cnidarian Ocelli and Supports the Presence of Photoisomerases
Erratum in
-
Corrigendum: Expression of opsins of the box jellyfish Tripedalia cystophora reveals the first photopigment in cnidarian ocelli and supports the presence of photoisomerases.Front Neuroanat. 2022 Sep 8;16:1028092. doi: 10.3389/fnana.2022.1028092. eCollection 2022. Front Neuroanat. 2022. PMID: 36157326 Free PMC article.
Abstract
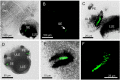
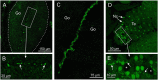
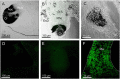
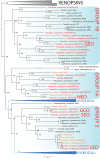
Cubomedusae, or box jellyfish, have a complex visual system comprising 24 eyes of four types. Like other cnidarians, their photoreceptor cells are ciliary in morphology, and a range of different techniques together show that at least two of the eye types-the image-forming upper and lower lens eyes-express opsin as the photopigment. The photoreceptors of these two eye types express the same opsin (Tc LEO), which belongs to the cnidarian-specific clade cnidops. Interestingly, molecular work has found a high number of opsin genes in box jellyfish, especially in the Caribbean species Tripedalia cystophora, most of which are of unknown function. In the current study, we raised antibodies against three out of five opsins identified from transcriptomic data from T. cystophora and used them to map the expression patterns. These expression patterns suggest one opsin as the photopigment in the slit eyes and another as a putative photoisomerase found in photoreceptors of all four eyes types. The last antibody stained nerve-like cells in the tentacles, in connection with nematocytes, and the radial nerve, in connection with the gonads. This is the first time photopigment expression has been localized to the outer segments of the photoreceptors in a cnidarian ocellus (simple eye). The potential presence of a photoisomerase could be another interesting convergence between box jellyfish and vertebrate photoreceptors, but it awaits final experimental proof.
Keywords: box jellyfish; cnidaria; cubozoa; opsin phylogeny; photopigment; phototransduction; vision.
Copyright © 2022 Garm, Svaerke, Pontieri and Oakley.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Corrigendum: Expression of opsins of the box jellyfish Tripedalia cystophora reveals the first photopigment in cnidarian ocelli and supports the presence of photoisomerases.Front Neuroanat. 2022 Sep 8;16:1028092. doi: 10.3389/fnana.2022.1028092. eCollection 2022. Front Neuroanat. 2022. PMID: 36157326 Free PMC article.
-
Immunohistochemical evidence for multiple photosystems in box jellyfish.Cell Tissue Res. 2008 Jul;333(1):115-24. doi: 10.1007/s00441-008-0614-8. Epub 2008 May 27. Cell Tissue Res. 2008. PMID: 18504619
-
Ocular and extraocular expression of opsins in the rhopalium of Tripedalia cystophora (Cnidaria: Cubozoa).PLoS One. 2014 Jun 5;9(6):e98870. doi: 10.1371/journal.pone.0098870. eCollection 2014. PLoS One. 2014. PMID: 24901369 Free PMC article.
-
Simple Eyes, Extraocular Photoreceptors and Opsins in the American Horseshoe Crab.Integr Comp Biol. 2016 Nov;56(5):809-819. doi: 10.1093/icb/icw093. Epub 2016 Jul 21. Integr Comp Biol. 2016. PMID: 27444526 Review.
-
Evidence for multiple photosystems in jellyfish.Int Rev Cell Mol Biol. 2010;280:41-78. doi: 10.1016/S1937-6448(10)80002-4. Epub 2010 Mar 18. Int Rev Cell Mol Biol. 2010. PMID: 20797681 Review.
Cited by
-
Genome assembly of Bougainvillia cf. muscus (Cnidaria: Hydrozoa).G3 (Bethesda). 2025 Jul 9;15(7):jkaf110. doi: 10.1093/g3journal/jkaf110. G3 (Bethesda). 2025. PMID: 40388381 Free PMC article.
-
Phototactic preference and its genetic basis in the planulae of the colonial Hydrozoan Hydractinia symbiolongicarpus.bioRxiv [Preprint]. 2024 Apr 1:2024.03.28.585045. doi: 10.1101/2024.03.28.585045. bioRxiv. 2024. PMID: 38617216 Free PMC article. Preprint.
-
Comparative Analysis of Convergent Jellyfish Eyes Reveals Extensive Differences in Expression of Vision-Related Genes.Ecol Evol. 2025 Jul 14;15(7):e71784. doi: 10.1002/ece3.71784. eCollection 2025 Jul. Ecol Evol. 2025. PMID: 40661905 Free PMC article.
-
Deep Diversity: Extensive Variation in the Components of Complex Visual Systems across Animals.Cells. 2022 Dec 8;11(24):3966. doi: 10.3390/cells11243966. Cells. 2022. PMID: 36552730 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources

