Dimerization of GPCRs: Novel insight into the role of FLNA and SSAs regulating SST2 and SST5 homo- and hetero-dimer formation
- PMID: 35992099
- PMCID: PMC9389162
- DOI: 10.3389/fendo.2022.892668
Dimerization of GPCRs: Novel insight into the role of FLNA and SSAs regulating SST2 and SST5 homo- and hetero-dimer formation
Abstract
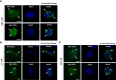
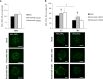
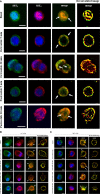
The process of GPCR dimerization can have profound effects on GPCR activation, signaling, and intracellular trafficking. Somatostatin receptors (SSTs) are class A GPCRs abundantly expressed in pituitary tumors where they represent the main pharmacological targets of somatostatin analogs (SSAs), thanks to their antisecretory and antiproliferative actions. The cytoskeletal protein filamin A (FLNA) directly interacts with both somatostatin receptor type 2 (SST2) and 5 (SST5) and regulates their expression and signaling in pituitary tumoral cells. So far, the existence and physiological relevance of SSTs homo- and hetero-dimerization in the pituitary have not been explored. Moreover, whether octreotide or pasireotide may play modulatory effects and whether FLNA may participate to this level of receptor organization have remained elusive. Here, we used a proximity ligation assay (PLA)-based approach for the in situ visualization and quantification of SST2/SST5 dimerization in rat GH3 as well as in human melanoma cells either expressing (A7) or lacking (M2) FLNA. First, we observed the formation of endogenous SST5 homo-dimers in GH3, A7, and M2 cells. Using the PLA approach combined with epitope tagging, we detected homo-dimers of human SST2 in GH3, A7, and M2 cells transiently co-expressing HA- and SNAP-tagged SST2. SST2 and SST5 can also form endogenous hetero-dimers in these cells. Interestingly, FLNA absence reduced the basal number of hetero-dimers (-36.8 ± 6.3% reduction of PLA events in M2, P < 0.05 vs. A7), and octreotide but not pasireotide promoted hetero-dimerization in both A7 and M2 (+20.0 ± 11.8% and +44.1 ± 16.3% increase of PLA events in A7 and M2, respectively, P < 0.05 vs. basal). Finally, immunofluorescence data showed that SST2 and SST5 recruitment at the plasma membrane and internalization are similarly induced by octreotide and pasireotide in GH3 and A7 cells. On the contrary, in M2 cells, octreotide failed to internalize both receptors whereas pasireotide promoted robust receptor internalization at shorter times than in A7 cells. In conclusion, we demonstrated that in GH3 cells SST2 and SST5 can form both homo- and hetero-dimers and that FLNA plays a role in the formation of SST2/SST5 hetero-dimers. Moreover, we showed that FLNA regulates SST2 and SST5 intracellular trafficking induced by octreotide and pasireotide.
Keywords: FLNA; GPCR dimerization; SST2; SST5; in situ PLA; somatostatin analogs.
Copyright © 2022 Treppiedi, Marra, Di Muro, Catalano, Mangili, Esposito, Calebiro, Arosio, Peverelli and Mantovani.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Filamin A is required for somatostatin receptor type 5 expression and pasireotide-mediated signaling in pituitary corticotroph tumor cells.Mol Cell Endocrinol. 2021 Mar 15;524:111159. doi: 10.1016/j.mce.2021.111159. Epub 2021 Jan 9. Mol Cell Endocrinol. 2021. PMID: 33428965
-
Cytoskeleton Protein Filamin A Is Required for Efficient Somatostatin Receptor Type 2 Internalization and Recycling through Rab5 and Rab4 Sorting Endosomes in Tumor Somatotroph Cells.Neuroendocrinology. 2020;110(7-8):642-652. doi: 10.1159/000503791. Epub 2019 Oct 1. Neuroendocrinology. 2020. PMID: 31574507
-
A novel pathway activated by somatostatin receptor type 2 (SST2): Inhibition of pituitary tumor cell migration and invasion through cytoskeleton protein recruitment.Int J Cancer. 2018 May 1;142(9):1842-1852. doi: 10.1002/ijc.31205. Epub 2017 Dec 20. Int J Cancer. 2018. PMID: 29226331
-
Somatostatin receptor expression and patients' response to targeted medical treatment in pituitary tumors: evidences and controversies.J Endocrinol Invest. 2020 Nov;43(11):1543-1553. doi: 10.1007/s40618-020-01335-0. Epub 2020 Jun 18. J Endocrinol Invest. 2020. PMID: 32557353 Review.
-
Cell specific interaction of pasireotide: review of preclinical studies in somatotroph and corticotroph pituitary cells.Pituitary. 2019 Feb;22(1):89-99. doi: 10.1007/s11102-018-0926-y. Pituitary. 2019. PMID: 30483918 Review.
Cited by
-
A supramolecular FRET signal amplification nanoprobe for high contrast and synchronous in situ imaging of cell surface receptor homodimers/heterodimers.Chem Sci. 2025 Feb 17;16(11):4732-4740. doi: 10.1039/d4sc08004a. eCollection 2025 Mar 12. Chem Sci. 2025. PMID: 39968283 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

