Impact of root hairs on microscale soil physical properties in the field
- PMID: 35992246
- PMCID: PMC9381483
- DOI: 10.1007/s11104-022-05530-1
Impact of root hairs on microscale soil physical properties in the field
Abstract
Aims: Recent laboratory studies revealed that root hairs may alter soil physical behaviour, influencing soil porosity and water retention on the small scale. However, the results are not consistent, and it is not known if structural changes at the small-scale have impacts at larger scales. Therefore, we evaluated the potential effects of root hairs on soil hydro-mechanical properties in the field using rhizosphere-scale physical measurements.
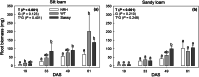
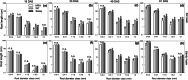
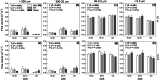
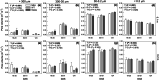
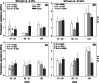
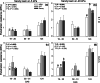
Methods: Changes in soil water retention properties as well as mechanical and hydraulic characteristics were monitored in both silt loam and sandy loam soils. Measurements were taken from plant establishment to harvesting in field trials, comparing three barley genotypes representing distinct phenotypic categories in relation to root hair length. Soil hardness and elasticity were measured using a 3-mm-diameter spherical indenter, while water sorptivity and repellency were measured using a miniaturized infiltrometer with a 0.4-mm tip radius.
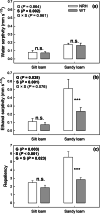
Results: Over the growing season, plants induced changes in the soil water retention properties, with the plant available water increasing by 21%. Both soil hardness (P = 0.031) and elasticity (P = 0.048) decreased significantly in the presence of root hairs in silt loam soil, by 50% and 36%, respectively. Root hairs also led to significantly smaller water repellency (P = 0.007) in sandy loam soil vegetated with the hairy genotype (-49%) compared to the hairless mutant.
Conclusions: Breeding of cash crops for improved soil conditions could be achieved by selecting root phenotypes that ameliorate soil physical properties and therefore contribute to increased soil health.
Supplementary information: The online version contains supplementary material available at 10.1007/s11104-022-05530-1.
Keywords: Barley; Root hairs; Soil health; Soil hydromechanical properties; Soil structure; Soil water retention.
© The Author(s) 2022.
Figures

Similar articles
-
Significance of root hairs for plant performance under contrasting field conditions and water deficit.Ann Bot. 2021 Jul 28;128(1):1-16. doi: 10.1093/aob/mcaa181. Ann Bot. 2021. PMID: 33038211 Free PMC article.
-
Plant influence on rhizosphere hydraulic properties: direct measurements using a miniaturized infiltrometer.New Phytol. 2003 Mar;157(3):597-603. doi: 10.1046/j.1469-8137.2003.00690.x. New Phytol. 2003. PMID: 33873394
-
Root hair length and rhizosheath mass depend on soil porosity, strength and water content in barley genotypes.Planta. 2014 Mar;239(3):643-51. doi: 10.1007/s00425-013-2002-1. Epub 2013 Dec 8. Planta. 2014. PMID: 24318401
-
Responses of root hairs to soil compaction: A review.Plant Sci. 2025 Jun;355:112461. doi: 10.1016/j.plantsci.2025.112461. Epub 2025 Mar 11. Plant Sci. 2025. PMID: 40081764 Review.
-
The role of root hairs in water uptake: recent advances and future perspectives.J Exp Bot. 2022 Jun 2;73(11):3330-3338. doi: 10.1093/jxb/erac114. J Exp Bot. 2022. PMID: 35323893 Review.
Cited by
-
Uncovering root compaction response mechanisms: new insights and opportunities.J Exp Bot. 2024 Jan 10;75(2):578-583. doi: 10.1093/jxb/erad389. J Exp Bot. 2024. PMID: 37950742 Free PMC article.
-
Germination Behavior and Geographical Information System-Based Phenotyping of Root Hairs to Evaluate the Effects of Different Sources of Black Soldier Fly (Hermetia illucens) Larval Frass on Herbaceous Crops.Plants (Basel). 2024 Jan 14;13(2):230. doi: 10.3390/plants13020230. Plants (Basel). 2024. PMID: 38256783 Free PMC article.
References
-
- Ahmed MA, Kroener E, Benard P, Zarebanadkouki M, Kaestner A, Carminati A. Drying of mucilage causes water repellency in the rhizosphere of maize: measurements and modelling. Plant Soil. 2016;407:161–171. doi: 10.1007/s11104-015-2749-1. - DOI
-
- Barré P, Hallett PD. Rheological stabilization of wet soils by model root and fungal exudates depends on clay mineralogy. Eur J Soil Sci. 2009;60:525–538. doi: 10.1111/j.1365-2389.2009.01151.x. - DOI
-
- Benard P, Kroener E, Vontobel P, Kaestner A, Carminati A. Water percolation through the root-soil interface. Adv Water Resour. 2016;95:190–198. doi: 10.1016/j.advwatres.2015.09.014. - DOI
-
- Bengough AG (2012) Water dynamics of the root zone: Rhizosphere biophysics and its control on soil hydrology. Vadose Zone J 11. 10.2136/vzj2011.0111
LinkOut - more resources
Full Text Sources
