Mutually Exclusive Interactions of Rifabutin with Spatially Distinct Mycobacterial Cell Envelope Membrane Layers Offer Insights into Membrane-Centric Therapy of Infectious Diseases
- PMID: 35996474
- PMCID: PMC9389580
- DOI: 10.1021/acsbiomedchemau.2c00010
Mutually Exclusive Interactions of Rifabutin with Spatially Distinct Mycobacterial Cell Envelope Membrane Layers Offer Insights into Membrane-Centric Therapy of Infectious Diseases
Abstract
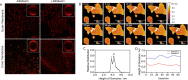
The mycobacterial cell envelope has spatially resolved inner and outer membrane layers with distinct compositions and membrane properties. However, the functional implication and relevance of this organization remain unknown. Using membrane biophysics and molecular simulations, we reveal a varied interaction profile of these layers with antibiotic Rifabutin, underlined by the structural and chemical makeup of the constituent lipids. The mycobacterial inner membrane displayed the highest partitioning of Rifabutin, which was located exclusively in the lipid head group/interfacial region. In contrast, the drug exhibited specific interaction sites in the head group/interfacial and hydrophobic acyl regions within the outer membrane. Altogether, we show that the design of membrane-active agents that selectively disrupt the mycobacterial outer membrane structure can increase drug uptake and enhance intracellular drug concentrations. Exploiting the mycobacterium-specific membrane-drug interaction profiles, chemotypes consisting of outer membrane-disruptive agents and antitubercular drugs can offer new opportunities for combinational tuberculosis (TB) therapy.
© 2022 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

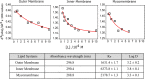
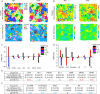
 (red circle) and dynamic quenching
(red circle) and dynamic quenching  (black square) of DPH and TMA–DPH
probes were measured against increasing concentrations of Rifabutin
(0–30 μM) in each Msm membrane system.
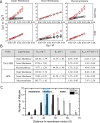
(B) Stern–Volmer quenching constant (KSV), dynamic quenching constant (KD), and bimolecular quenching constant (Kq) were derived from the static and lifetime fluorescence plots to
determine the extent of quenching by, and the accessibility of, Rifabutin
to DPH/TMA–DPH fluorescence probes embedded in different Msm membrane systems, respectively. (C) Number of Rifabutin
molecules in the regions at different z-direction
distances from the membrane center.
(black square) of DPH and TMA–DPH
probes were measured against increasing concentrations of Rifabutin
(0–30 μM) in each Msm membrane system.
(B) Stern–Volmer quenching constant (KSV), dynamic quenching constant (KD), and bimolecular quenching constant (Kq) were derived from the static and lifetime fluorescence plots to
determine the extent of quenching by, and the accessibility of, Rifabutin
to DPH/TMA–DPH fluorescence probes embedded in different Msm membrane systems, respectively. (C) Number of Rifabutin
molecules in the regions at different z-direction
distances from the membrane center.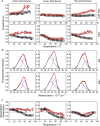
Similar articles
-
Biophysical characterization of mycobacterial model membranes and their interaction with rifabutin: Towards lipid-guided drug screening in tuberculosis.Biochim Biophys Acta Biomembr. 2019 Jun 1;1861(6):1213-1227. doi: 10.1016/j.bbamem.2019.04.004. Epub 2019 Apr 17. Biochim Biophys Acta Biomembr. 2019. PMID: 31002767
-
Lipid Clustering in Mycobacterial Cell Envelope Layers Governs Spatially Resolved Solvation Dynamics.Chem Asian J. 2022 Jun 1;17(11):e202200146. doi: 10.1002/asia.202200146. Epub 2022 Apr 13. Chem Asian J. 2022. PMID: 35419975
-
[Development of antituberculous drugs: current status and future prospects].Kekkaku. 2006 Dec;81(12):753-74. Kekkaku. 2006. PMID: 17240921 Review. Japanese.
-
Fusion Landscape of Mycobacterial Envelope-Derived Lipid Vesicles with Intact Bacteria Dictates High Intracellular Drug Retention.ACS Appl Bio Mater. 2023 Aug 21;6(8):3066-3073. doi: 10.1021/acsabm.3c00286. Epub 2023 Jul 26. ACS Appl Bio Mater. 2023. PMID: 37493278
-
Mycobacterial Membranes as Actionable Targets for Lipid-Centric Therapy in Tuberculosis.J Med Chem. 2022 Feb 24;65(4):3046-3065. doi: 10.1021/acs.jmedchem.1c01870. Epub 2022 Feb 8. J Med Chem. 2022. PMID: 35133820 Review.
Cited by
-
Mycobacterial lipid-derived immunomodulatory drug- liposome conjugate eradicates endosome-localized mycobacteria.J Control Release. 2023 Aug;360:578-590. doi: 10.1016/j.jconrel.2023.07.013. Epub 2023 Jul 14. J Control Release. 2023. PMID: 37442202 Free PMC article.
-
Decoding the role of mycobacterial lipid remodelling and membrane dynamics in antibiotic tolerance.Chem Sci. 2024 Oct 29;15(45):19084-93. doi: 10.1039/d4sc06618a. Online ahead of print. Chem Sci. 2024. PMID: 39483253 Free PMC article.
-
Effect of Host Cholesterol on the Membrane Dynamics of Outer Membrane Lipids of Mycobacteria.Chem Asian J. 2023 Dec 1;18(23):e202300697. doi: 10.1002/asia.202300697. Epub 2023 Oct 27. Chem Asian J. 2023. PMID: 37846643 Free PMC article.
-
Temperature-Induced Restructuring of Mycolic Acid Bilayers Modeling the Mycobacterium tuberculosis Outer Membrane: A Molecular Dynamics Study.Molecules. 2024 Feb 2;29(3):696. doi: 10.3390/molecules29030696. Molecules. 2024. PMID: 38338443 Free PMC article.
References
-
- WHO . Global Tuberculosis Report 2020; World Health Organization: Geneva, 2020. https://www.who.int/publications/i/item/9789240013131.
LinkOut - more resources
Full Text Sources

