Antithrombotic Therapy for Symptomatic Peripheral Arterial Disease: A Systematic Review and Network Meta-Analysis
- PMID: 35997941
- PMCID: PMC9499921
- DOI: 10.1007/s40265-022-01756-6
Antithrombotic Therapy for Symptomatic Peripheral Arterial Disease: A Systematic Review and Network Meta-Analysis
Erratum in
-
Correction to: Antithrombotic Therapy for Symptomatic Peripheral Arterial Disease: A Systematic Review and Network Meta-Analysis.Drugs. 2022 Sep;82(13):1435. doi: 10.1007/s40265-022-01776-2. Drugs. 2022. PMID: 36070076 Free PMC article. No abstract available.
Abstract
Background: High-quality evidence from trials directly comparing single antiplatelet therapies in symptomatic peripheral arterial disease (PAD) to dual antiplatelet therapies or acetylsalicylic acid (ASA) plus low-dose rivaroxaban is lacking. Therefore, we conducted a network meta-analysis on the effectiveness of all antithrombotic regimens studied in PAD.
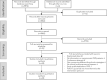
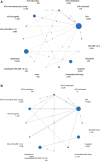
Methods: A systematic search was conducted to identify randomized controlled trials. The primary endpoints were major adverse cardiovascular events (MACE) and major bleedings. Secondary endpoints were major adverse limb events (MALE) and acute limb ischaemia (ALI). For each outcome, a frequentist network meta-analysis was used to compare relative risks (RRs) between medication and ASA. ASA was the universal comparator since a majority of studies used ASA as in the reference group.
Results: Twenty-four randomized controlled trials were identified including 48,759 patients. With regard to reducing MACE, clopidogrel [RR 0.78, 95% confidence interval (CI) 0.66-0.93], ticagrelor (RR 0.79, 95% CI 0.65-0.97), ASA plus ticagrelor (RR 0.79, 95% CI 0.64-0.97), and ASA plus low-dose rivaroxaban (RR 0.84, 95% CI 0.76-0.93) were more effective than ASA, and equally effective to one another. As compared to ASA, major bleedings occurred more frequently with vitamin K antagonists, rivaroxaban, ASA plus vitamin K antagonists, and ASA plus low-dose rivaroxaban. All regimens were similar to ASA concerning MALE, while ASA plus low-dose rivaroxaban was more effective in preventing ALI (RR 0.67, 95% CI 0.55-0.80). Subgroup analysis in patients undergoing peripheral revascularization revealed that ≥ 3 months after intervention, evidence of benefit regarding clopidogrel, ticagrelor, and ASA plus ticagrelor was lacking, while ASA plus low-dose rivaroxaban was more effective in preventing MACE (RR 0.87, 95% CI 0.78-0.97) and MALE (RR 0.89, 95% CI 0.81-0.97) compared to ASA. ASA plus clopidogrel was not superior to ASA in preventing MACE ≥ 3 months after revascularization. Evidence regarding antithrombotic treatment strategies within 3 months after a peripheral intervention was lacking.
Conclusion: Clopidogrel, ticagrelor, ASA plus ticagrelor, and ASA plus low-dose rivaroxaban are superior to ASA monotherapy and equally effective to one another in preventing MACE in PAD. Of these four therapies, only ASA plus low-dose rivaroxaban provides a higher risk of major bleedings. More than 3 months after peripheral vascular intervention, ASA plus low-dose rivaroxaban is superior in preventing MACE and MALE compared to ASA but again at the cost of a higher risk of bleeding, while other treatment regimens show non-superiority. Based on the current evidence, clopidogrel may be considered the antithrombotic therapy of choice for most PAD patients, while in patients who underwent a peripheral vascular intervention, ASA plus low-dose rivaroxaban could be considered for the long-term (> 3 months) prevention of MACE and MALE.
© 2022. The Author(s).
Conflict of interest statement
Loes H. Willems, Dominique P.M.S.M. Maas, Kees Kramers, Michel M.P.J. Reijnen, Niels P. Riksen, Hugo Ten Cate, Rozemarijn J. van der Vijver-Coppen, Gert J. de Borst, Barend M. E. Mees, Clark J. Zeebregts, Gerjon Hannink, and Michiel C. Warlé declare no relevant financial or non-financial interests.
Figures

Similar articles
-
Antiplatelet and anticoagulant drugs for prevention of restenosis/reocclusion following peripheral endovascular treatment.Cochrane Database Syst Rev. 2012 Aug 15;2012(8):CD002071. doi: 10.1002/14651858.CD002071.pub3. Cochrane Database Syst Rev. 2012. PMID: 22895926 Free PMC article.
-
Drugs for preventing postoperative nausea and vomiting in adults after general anaesthesia: a network meta-analysis.Cochrane Database Syst Rev. 2020 Oct 19;10(10):CD012859. doi: 10.1002/14651858.CD012859.pub2. Cochrane Database Syst Rev. 2020. PMID: 33075160 Free PMC article.
-
Prophylactic Efficacy and Safety of Antithrombotic Regimens in Patients with Stable Atherosclerotic Cardiovascular Disease (S-ASCVD): A Bayesian Network Meta-Regression Analysis.Am J Cardiovasc Drugs. 2023 May;23(3):257-267. doi: 10.1007/s40256-023-00574-9. Epub 2023 Mar 3. Am J Cardiovasc Drugs. 2023. PMID: 36867384
-
Clinical effectiveness and cost-effectiveness of clopidogrel and modified-release dipyridamole in the secondary prevention of occlusive vascular events: a systematic review and economic evaluation.Health Technol Assess. 2004 Oct;8(38):iii-iv, 1-196. doi: 10.3310/hta8380. Health Technol Assess. 2004. PMID: 15461876
-
Antiplatelet agents and anticoagulants for hypertension.Cochrane Database Syst Rev. 2022 Jul 28;7(7):CD003186. doi: 10.1002/14651858.CD003186.pub4. Cochrane Database Syst Rev. 2022. PMID: 35900898 Free PMC article.
Cited by
-
Integrating Mechanisms in Thrombotic Peripheral Arterial Disease.Pharmaceuticals (Basel). 2022 Nov 18;15(11):1428. doi: 10.3390/ph15111428. Pharmaceuticals (Basel). 2022. PMID: 36422558 Free PMC article. Review.
-
Antithrombotic therapy following lower extremity endovascular revascularization: The results of a survey of vascular specialists.JVS Vasc Insights. 2024;2:100153. doi: 10.1016/j.jvsvi.2024.100153. Epub 2024 Oct 28. JVS Vasc Insights. 2024. PMID: 39877294 Free PMC article.
-
L-theanine promotes angiogenesis in limb ischemic mice by modulating NRP1/VEGFR2 signaling.Biomol Biomed. 2025 Apr 3;25(5):1063-1078. doi: 10.17305/bb.2024.11256. Biomol Biomed. 2025. PMID: 39803888 Free PMC article.
-
Impact of CYP2C19 Genotype Status on Clinical Outcomes in Patients with Symptomatic Coronary Artery Disease, Stroke, and Peripheral Arterial Disease: A Systematic Review and Meta-Analysis.Drugs. 2024 Oct;84(10):1275-1297. doi: 10.1007/s40265-024-02076-7. Epub 2024 Sep 5. Drugs. 2024. PMID: 39235670 Free PMC article.
-
Medication adherence of patients with peripheral arterial disease to antithrombotic therapy: a systematic review.BMJ Open. 2025 Feb 22;15(2):e085056. doi: 10.1136/bmjopen-2024-085056. BMJ Open. 2025. PMID: 39987003 Free PMC article.
References
-
- Aboyans V, Ricco JB, Bartelink MEL et al. 2017 ESC Guidelines on the Diagnosis and Treatment of Peripheral Arterial Diseases, in collaboration with the European Society for Vascular Surgery (ESVS): Document covering atherosclerotic disease of extracranial carotid and vertebral, mesenteric, renal, upper and lower extremity arteries. Endorsed by: the European Stroke Organization (ESO)The Task Force for the Diagnosis and Treatment of Peripheral Arterial Diseases of the European Society of Cardiology (ESC) and of the European Society for Vascular Surgery (ESVS). Eur Heart J. 2018;39(9):763–816. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

