Next-generation transcatheter aortic valve implantation
- PMID: 36003870
- PMCID: PMC9390526
- DOI: 10.1016/j.xjon.2020.06.008
Next-generation transcatheter aortic valve implantation
Abstract
Objective: Transcatheter aortic valve implantation (TAVI) procedures are increasing rapidly, but the durability of tissue valve and periprocedural complications are not satisfactory. Immune reaction to the galactose-α-1,3 galactose β-1,4-N-acetylglucosamine (α-Gal) and conventional processing protocols of cardiac xenografts lead to calcification. Next-generation TAVI needs to be made with α-Gal-free xenografts by multiple anticalcification therapies to avoid immune rejection and enhance durability, and three-dimensional (3D) printing technology to improve the procedural safety.
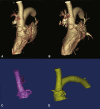
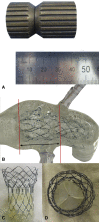
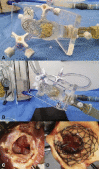
Methods: Porcine pericardia were decellularized and immunologically modified with α-galactosidase. The pericardia were treated by space filler, crosslinked with glutaraldehyde in organic solvent, and detoxified. The sheep-specific nitinol (nickel-titanium memory alloy) wire backbone was made from a 3D-printed model for ovine aortic root. After it passed the fitting test, we manufactured a self-expandable stented valve with the porcine pericardia mounted on the customized nitinol wire-based stent. After in vitro circulation using customized silicone aortic root, we performed TAVI in 9 sheep and obtained hemodynamic, radiological, immunohistopathological, and biochemical results.
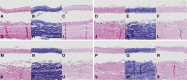
Results: The valve functioned well, with excellent stent fitting and good coronary flow under in vitro circulation. Sheep were sequentially scheduled to be humanely killed until 238 days after TAVI. Echocardiography and cardiac catheterization demonstrated good hemodynamic status and function of the aortic valve. The xenografts were well preserved without α-Gal immune reaction or calcification based on the immunological, radiographic, microscopic, and biochemical examinations.
Conclusions: We proved preclinical safety and efficacy for next-generation α-Gal-free TAVI with multiple anticalcification therapies and 3D-printing technology. A future clinical study is warranted based on these promising preclinical results.
Keywords: 3D, 3-dimensional; BSA, bovine serum albumin; GA, glutaraldehyde; TAVI, transcatheter aortic valve implantation; bioengineering; biomaterials; bioprosthesis; calcification; heart valve; xenograft; α-Gal KO, α1,3-galactosyltransferase knockout; α-Gal, galactose-α-1,3 galactose β-1,4-N-acetylglucosamine.
© 2020 The Authors.
Figures


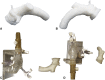



Similar articles
-
Development of a next-generation tissue valve using a glutaraldehyde-fixed porcine aortic valve treated with decellularization, α-galactosidase, space filler, organic solvent and detoxification.Eur J Cardiothorac Surg. 2015 Jul;48(1):104-13. doi: 10.1093/ejcts/ezu385. Epub 2014 Oct 14. Eur J Cardiothorac Surg. 2015. PMID: 25315752
-
A preclinical trial of perventricular pulmonary valve implantation: Pericardial versus aortic porcine valves mounted on self-expandable stent.Artif Organs. 2021 May;45(5):E89-E100. doi: 10.1111/aor.13845. Epub 2020 Dec 6. Artif Organs. 2021. PMID: 33090503
-
Valved conduit with glutaraldehyde-fixed bovine pericardium treated by anticalcification protocol.Korean J Thorac Cardiovasc Surg. 2014 Aug;47(4):333-43. doi: 10.5090/kjtcs.2014.47.4.333. Epub 2014 Aug 5. Korean J Thorac Cardiovasc Surg. 2014. PMID: 25207241 Free PMC article.
-
Tissue engineered heart valves for transcatheter aortic valve implantation: current state, challenges, and future developments.Expert Rev Cardiovasc Ther. 2020 Oct;18(10):681-696. doi: 10.1080/14779072.2020.1792777. Epub 2020 Sep 23. Expert Rev Cardiovasc Ther. 2020. PMID: 32791869 Review.
-
Valve durability after transcatheter aortic valve implantation.J Thorac Dis. 2018 Nov;10(Suppl 30):S3629-S3636. doi: 10.21037/jtd.2018.07.38. J Thorac Dis. 2018. PMID: 30505545 Free PMC article. Review.
Cited by
-
Commentary: Rainmaking the "next-generation transcatheter aortic valve implantation" by anointing multidisciplinary perceptions.JTCVS Open. 2020 Jul 7;3:25-26. doi: 10.1016/j.xjon.2020.07.001. eCollection 2020 Sep. JTCVS Open. 2020. PMID: 36003858 Free PMC article. No abstract available.
-
Clinical Results for Cardiovascular Xenografts Treated With Novel Anticalcification Protocols.Artif Organs. 2025 Aug;49(8):1307-1313. doi: 10.1111/aor.15010. Epub 2025 Apr 11. Artif Organs. 2025. PMID: 40213904 Free PMC article.
References
-
- Rahhab Z., El Faquir N., Tchetche D., Delgado V., Kodali S., Mara Vollema E., et al. Expanding the indications for transcatheter aortic valve implantation. Nat Rev Cardiol. 2020;17:75–84. - PubMed
-
- Van Belle E., Vincent F., Labreuche J., Auffret V., Debry N., Lefèvre T., et al. Balloon-expandable versus self-expanding transcatheter aortic valve replacement: a propensity-matched comparison from the FRANCE-TAVI Registry. Circulation. 2020;141:243–259. - PubMed
-
- Blackman D.J., Saraf S., MacCarthy P.A., Myat A., Anderson S.G., Malkin C.J., et al. Long-term durability of transcatheter aortic valve prostheses. J Am Coll Cardiol. 2019;73:537–545. - PubMed
-
- Rheude T., Pellegrini C., Cassese S., Wiebe J., Wagner S., Trenkwalder T., et al. Predictors of haemodynamic structural valve deterioration following transcatheter aortic valve implantation with latest-generation balloon-expandable valves. EuroIntervention. 2020;15:1233–1239. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

