Computer simulations of transapical mitral valve repair with neochordae implantation: Clinical implications
- PMID: 36003874
- PMCID: PMC9390497
- DOI: 10.1016/j.xjon.2020.05.010
Computer simulations of transapical mitral valve repair with neochordae implantation: Clinical implications
Abstract
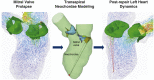
Objectives: Transapical beating heart neochordae implantation is an innovative mitral valve repair technique that has demonstrated promising clinical results in patients with primary mitral regurgitation. However, as clinical experience continues to increase, neochordae implantation criteria have not been fully standardized. The aim of this study was to investigate the biomechanical effects of selecting an antero-lateral apical access site compared with a postero-lateral site, and suboptimal neochordae length compared with optimal suture length, on restoring physiologic left heart dynamics.
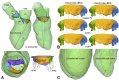
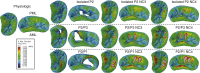
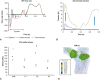
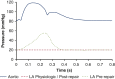
Methods: Transapical neochordae implantation using 3 and 4 sutures was computer simulated under 3 posterior mitral leaflet prolapse conditions: isolated P2, multiscallop P2/P3 and multiscallop P2/P1. Physiologic, pre- and postrepair left heart dynamics were evaluated using a fluid-structure interaction modeling framework.
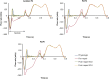
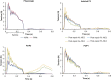
Results: Despite the absence of residual mitral regurgitation in all postrepair models with optimal neochordae length, selecting an antero-lateral apical entry site for the treatment of P2/P3 prolapse generated a significant increase (>80%) in neochordae tension and P2 peak stress, with respect to a postero-lateral entry site. During isolated P2 prolapse repair, although neochordae overtension by 5% led to minimal hemodynamic changes in the regurgitant volume compared with using an optimal suture length, a significant increase in systolic and diastolic neochordae tension (>300%) and posterior leaflet average stress (70%-460%) was quantified. On the other hand, neochordae undertension by 5% led to worsening of regurgitation severity.
Conclusions: This parametric computer study represents a further step toward an improved understanding of the biomechanical outcomes of transapical neochordae technologies.
Keywords: AL-NC, antero-lateral neochordae; AML, anterior mitral leaflet; AV, aortic valve; FSI, fluid-structure interaction; LV, left ventricle; MR, mitral regurgitation; MV, mitral valve; NC, neochordae; PL-NC, postero-lateral neochordae; PM, papillary muscle; PML, posterior mitral leaflet; beating heart mitral valve repair; ePTFE suture; ePTFE, expanded polytetrafluoroethylene; fluid-structure interaction FSI; primary mitral regurgitation; transapical neochord.
© 2020 The Authors.
Figures




Similar articles
-
Transapical mitral valve repair with neochordae implantation: FSI analysis of neochordae number and complexity of leaflet prolapse.Int J Numer Method Biomed Eng. 2020 Mar;36(3):e3297. doi: 10.1002/cnm.3297. Epub 2019 Dec 20. Int J Numer Method Biomed Eng. 2020. PMID: 31833663
-
Measuring chordae tension during transapical neochordae implantation: Toward understanding objective consequences of mitral valve repair.J Thorac Cardiovasc Surg. 2019 Sep;158(3):746-755. doi: 10.1016/j.jtcvs.2018.10.029. Epub 2018 Oct 17. J Thorac Cardiovasc Surg. 2019. PMID: 30454983
-
Good 5-Year Durability of Transapical Beating Heart Off-Pump Mitral Valve Repair With Neochordae.Ann Thorac Surg. 2018 Aug;106(2):440-445. doi: 10.1016/j.athoracsur.2018.01.092. Epub 2018 May 2. Ann Thorac Surg. 2018. PMID: 29729267
-
Microinvasive mitral valve repair with transapical mitral neochordae implantation.Front Cardiovasc Med. 2023 Jul 28;10:1166892. doi: 10.3389/fcvm.2023.1166892. eCollection 2023. Front Cardiovasc Med. 2023. PMID: 37576109 Free PMC article. Review.
-
Transapical NeoChord mitral valve repair.Ann Cardiothorac Surg. 2018 Nov;7(6):812-820. doi: 10.21037/acs.2018.11.04. Ann Cardiothorac Surg. 2018. PMID: 30598897 Free PMC article. Review.
Cited by
-
A CFD study on the interplay of torsion and vortex guidance by the mitral valve on the left ventricular wash-out making use of overset meshes (Chimera technique).Front Med Technol. 2022 Dec 22;4:1018058. doi: 10.3389/fmedt.2022.1018058. eCollection 2022. Front Med Technol. 2022. PMID: 36619345 Free PMC article.
-
Commentary: Neochord integrity: More than just initial breaking strength.JTCVS Open. 2021 Aug 12;8:276-277. doi: 10.1016/j.xjon.2021.08.007. eCollection 2021 Dec. JTCVS Open. 2021. PMID: 36004200 Free PMC article. No abstract available.
-
Commentary: Computational approaches for valve repair: Calculating the unknown.JTCVS Open. 2020 Jun 20;3:46-47. doi: 10.1016/j.xjon.2020.06.005. eCollection 2020 Sep. JTCVS Open. 2020. PMID: 36003859 Free PMC article. No abstract available.
-
Commentary: Mitral valve neo-chords, still a lot to learn!JTCVS Open. 2020 Jun 27;3:45. doi: 10.1016/j.xjon.2020.06.009. eCollection 2020 Sep. JTCVS Open. 2020. PMID: 36003877 Free PMC article. No abstract available.
References
-
- Gabbay U., Yosefy C. The underlying causes of chordae tendinae rupture: a systematic review. Int J Cardiol. 2010;143:113–118. - PubMed
-
- Espiritu D., Onohara D., Kalra K., Sarin E.L., Padala M. Transcatheter mitral valve repair therapies: evolution, status and challenges. Ann Biomed Eng. 2017;45:332–359. - PubMed
-
- Colli A., Bizzotto E., Manzan E., Besola L., Pradegan N., Bellu R., et al. Patient-specific ventricular access site selection for the neochord mitral valve repair procedure. Ann Thorac Surg. 2017;104:e199–e202. - PubMed
LinkOut - more resources
Full Text Sources

