Metastatic SDH-Deficient GIST Diagnosed during Pregnancy: Approach to a Complex Case
- PMID: 36005206
- PMCID: PMC9406627
- DOI: 10.3390/curroncol29080468
Metastatic SDH-Deficient GIST Diagnosed during Pregnancy: Approach to a Complex Case
Abstract
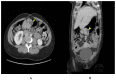
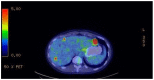
Gastrointestinal stromal tumors (GISTs) account for 1% of GI neoplasms in adults, and epidemiological data suggest an even lower occurrence in pregnant women. The majority of GISTs are caused by KIT and PDGFRA mutations. This is not the case in women of childbearing age. Some GISTs do not have a KIT/PDGFRA mutation and are classified as wild-type (WT) GISTs. WT-GIST includes many molecular subtypes including SDH deficiencies. In this paper, we present the first case report of a metastatic SDH-deficient GIST in a 23-year-old pregnant patient and the challenges encountered given her concurrent pregnancy. Our patient underwent a surgical tumor resection of her gastric GIST as well as a lymphadenectomy a week after induction of labor at 37 + 1 weeks. She received imatinib, sunitinib as well as regorafenib afterward. These drugs were discontinued because of disease progression despite treatment or after side effects were reported. Hence, she is currently under treatment with ripretinib. Her last FDG-PET showed a stable disease. This case highlights the complexity of GI malignancy care during pregnancy, and the presentation and management particularities of metastatic WT-GISTs. This case also emphasizes the need for a multidisciplinary approach and better clinical guidelines for offering optimal management to women in this specific context.
Keywords: GIST; SDH deficient; abdominal mass; gastrointestinal stromal tumor; malignancy; pregnancy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Gastrointestinal stromal tumors - Summary of mutational status of the primary/secondary KIT/PDGFRA mutations, BRAF mutations and SDH defects.Pathol Res Pract. 2019 Dec;215(12):152708. doi: 10.1016/j.prp.2019.152708. Epub 2019 Oct 29. Pathol Res Pract. 2019. PMID: 31708372
-
Identifying and managing rare subtypes of gastrointestinal stromal tumors.Expert Rev Gastroenterol Hepatol. 2025 Apr-May;19(5):549-561. doi: 10.1080/17474124.2025.2486304. Epub 2025 Apr 1. Expert Rev Gastroenterol Hepatol. 2025. PMID: 40156874 Review.
-
[Gastrointestinal stromal tumors (GIST)].Wien Med Wochenschr. 2023 Jun;173(9-10):201-205. doi: 10.1007/s10354-022-00965-8. Epub 2022 Sep 26. Wien Med Wochenschr. 2023. PMID: 36155864 German.
-
Molecular Subtypes of KIT/PDGFRA Wild-Type Gastrointestinal Stromal Tumors: A Report From the National Institutes of Health Gastrointestinal Stromal Tumor Clinic.JAMA Oncol. 2016 Jul 1;2(7):922-8. doi: 10.1001/jamaoncol.2016.0256. JAMA Oncol. 2016. PMID: 27011036 Free PMC article.
-
Succinate dehydrogenase deficiency in a PDGFRA mutated GIST.BMC Cancer. 2017 Aug 2;17(1):512. doi: 10.1186/s12885-017-3499-7. BMC Cancer. 2017. PMID: 28768491 Free PMC article. Review.
Cited by
-
Diagnosis and management of high risk gastrointestinal stromal tumor in first trimester pregnancy: A case report and review of the literature.Heliyon. 2023 May 9;9(5):e15858. doi: 10.1016/j.heliyon.2023.e15858. eCollection 2023 May. Heliyon. 2023. PMID: 37215772 Free PMC article.
References
-
- Demetri G.D., von Mehren M., Blanke C.D., Van den Abbeele A.D., Eisenberg B., Roberts P.J., Heinrich M.J., Tuveson D.J., Singer S., Janicek M., et al. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N. Engl. J. Med. 2002;347:472–480. doi: 10.1056/NEJMoa020461. - DOI - PubMed
-
- Joensuu H., Eriksson M., Hall K.S., Hartmann J.T., Pink D., Schütte J., Ramadori G., Hohenberger P., Duyster J., Al-Batran S.-E., et al. One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor: A randomized trial. JAMA. 2012;307:1265–1272. doi: 10.1001/jama.2012.347. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

