Pyrethroids in an AlphaFold2 Model of the Insect Sodium Channel
- PMID: 36005370
- PMCID: PMC9409284
- DOI: 10.3390/insects13080745
Pyrethroids in an AlphaFold2 Model of the Insect Sodium Channel
Abstract
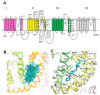
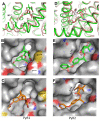
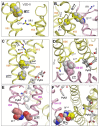
Pyrethroid insecticides stabilize the open state of insect sodium channels. Previous mutational, electrophysiological, and computational analyses led to the development of homology models predicting two pyrethroid receptor sites, PyR1 and PyR2. Many of the naturally occurring sodium channel mutations, which confer knockdown resistance (kdr) to pyrethroids, are located within or close to these receptor sites, indicating that these mutations impair pyrethroid binding. However, the mechanism of the state-dependent action of pyrethroids and the mechanisms by which kdr mutations beyond the receptor sites confer resistance remain unclear. Recent advances in protein structure prediction using the AlphaFold2 (AF2) neural network allowed us to generate a new model of the mosquito sodium channel AaNav1-1, with the activated voltage-sensing domains (VSMs) and the presumably inactivated pore domain (PM). We further employed Monte Carlo energy minimizations to open PM and deactivate VSM-I and VSM-II to generate additional models. The docking of a Type II pyrethroid deltamethrin in the models predicted its interactions with many known pyrethroid-sensing residues in the PyR1 and PyR2 sites and revealed ligand-channel interactions that stabilized the open PM and activated VSMs. Our study confirms the predicted two pyrethroid receptor sites, explains the state-dependent action of pyrethroids, and proposes the mechanisms of the allosteric effects of various kdr mutations on pyrethroid action. The AF2-based models may assist in the structure-based design of new insecticides.
Keywords: channel gating; insecticides; kdr mutations; pyrethroids; sodium channel.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

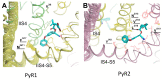
Similar articles
-
Rotational Symmetry of Two Pyrethroid Receptor Sites in the Mosquito Sodium Channel.Mol Pharmacol. 2015 Aug;88(2):273-80. doi: 10.1124/mol.115.098707. Epub 2015 May 13. Mol Pharmacol. 2015. PMID: 25972447 Free PMC article.
-
Characterization of two kdr mutations at predicted pyrethroid receptor site 2 in the sodium channels of Aedes aegypti and Nilaparvata lugens.Insect Biochem Mol Biol. 2022 Sep;148:103814. doi: 10.1016/j.ibmb.2022.103814. Epub 2022 Aug 3. Insect Biochem Mol Biol. 2022. PMID: 35932971 Free PMC article.
-
A unique mechanism of transfluthrin action revealed by mapping its binding sites in the mosquito sodium channel.Insect Biochem Mol Biol. 2024 Dec;175:104214. doi: 10.1016/j.ibmb.2024.104214. Epub 2024 Nov 19. Insect Biochem Mol Biol. 2024. PMID: 39566664
-
The molecular interactions of pyrethroid insecticides with insect and mammalian sodium channels.Pest Manag Sci. 2001 Oct;57(10):877-88. doi: 10.1002/ps.392. Pest Manag Sci. 2001. PMID: 11695180 Review.
-
Mutations in the voltage-gated sodium channel gene of anophelines and their association with resistance to pyrethroids - a review.Parasit Vectors. 2014 Oct 7;7:450. doi: 10.1186/1756-3305-7-450. Parasit Vectors. 2014. PMID: 25292318 Free PMC article. Review.
Cited by
-
First report on knockdown resistance mutations in wild populations of Aedes aegypti from Argentina determined by a novel multiplex high-resolution melting polymerase chain reaction method.Parasit Vectors. 2023 Jul 6;16(1):222. doi: 10.1186/s13071-023-05840-y. Parasit Vectors. 2023. PMID: 37415215 Free PMC article.
-
AlphaFold2 and its applications in the fields of biology and medicine.Signal Transduct Target Ther. 2023 Mar 14;8(1):115. doi: 10.1038/s41392-023-01381-z. Signal Transduct Target Ther. 2023. PMID: 36918529 Free PMC article. Review.
-
Discovery of novel acetylcholinesterase inhibitors through AI-powered structure prediction and high-performance computing-enhanced virtual screening.RSC Adv. 2025 Feb 7;15(6):4262-4273. doi: 10.1039/d4ra07951e. eCollection 2025 Feb 6. RSC Adv. 2025. PMID: 39926230 Free PMC article.
-
Bridging prediction and reality: Comprehensive analysis of experimental and AlphaFold 2 full-length nuclear receptor structures.Comput Struct Biotechnol J. 2025 May 15;27:1998-2013. doi: 10.1016/j.csbj.2025.05.010. eCollection 2025. Comput Struct Biotechnol J. 2025. PMID: 40496892 Free PMC article.
-
Acaricide Resistance Monitoring and Structural Insights for Precision Tetranychus urticae Management.Insects. 2025 Apr 23;16(5):440. doi: 10.3390/insects16050440. Insects. 2025. PMID: 40429153 Free PMC article.
References
-
- Narahashi T. Mechanisms of action of pyretrhoids on sodium and calcium channel gating. Neuropharmacol. Pestic. Action. 1986:36–60.
Grants and funding
LinkOut - more resources
Full Text Sources

