Discovery of Ircinianin Lactones B and C-Two New Cyclic Sesterterpenes from the Marine Sponge Ircinia wistarii
- PMID: 36005535
- PMCID: PMC9410537
- DOI: 10.3390/md20080532
Discovery of Ircinianin Lactones B and C-Two New Cyclic Sesterterpenes from the Marine Sponge Ircinia wistarii
Abstract
Two new ircinianin-type sesterterpenoids, ircinianin lactone B and ircinianin lactone C (7 and 8), together with five known entities from the ircinianin compound family (1, 3-6) were isolated from the marine sponge Ircinia wistarii. Ircinianin lactones B and C (7 and 8) represent new ircinianin terpenoids with a modified oxidation pattern. Despite their labile nature, the structures could be established using a combination of spectroscopic data, including HRESIMS and 1D/2D NMR techniques, as well as computational chemistry and quantum-mechanical calculations. In a broad screening approach for biological activity, the class-defining compound ircinianin (1) showed moderate antiprotozoal activity against Plasmodium falciparum (IC50 25.4 μM) and Leishmania donovani (IC50 16.6 μM).
Keywords: Ircinia wistarii; Leishmania donovani; NCI-60; Plasmodium falciparum; antiprotozoal activity; bioactivity screening; ircinianin-derivates; sesterterpene; sponge.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

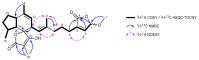
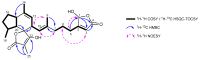
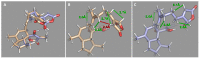
References
-
- Manes L.V., Naylor S., Crews P., Bakus G.J. Suvanine, a novel sesterterpene from an Ircinia marine sponge. J. Org. Chem. 1985;50:284–286. doi: 10.1021/jo00202a030. - DOI
-
- Höller U., König G.M., Wright A.D. Two New Sesterterpene Tetronic Acids from the Marine Sponge Ircinia oros. J. Nat. Prod. 1997;60:832–835. doi: 10.1021/np970170y. - DOI
-
- Wright A.E., McCarthy P.J., Schulte G.K. Sulfircin: A new sesterterpene sulfate from a deep-water sponge of the genus Ircinia. J. Org. Chem. 1989;54:3472–3474. doi: 10.1021/jo00275a037. - DOI
-
- Barrow C.J., Blunt J.W., Munro M.H.G., Perry N.B. Oxygenated Furanosesterterpene Tetronic Acids from a Sponge of the Genus Ircinia. J. Nat. Prod. 1988;51:1294–1298. doi: 10.1021/np50060a047. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

