The "Bloodless" Blood Test: Intradermal Prick Nanoelectronics for the Blood Extraction-Free Multiplex Detection of Protein Biomarkers
- PMID: 36006419
- PMCID: PMC9527802
- DOI: 10.1021/acsnano.2c01793
The "Bloodless" Blood Test: Intradermal Prick Nanoelectronics for the Blood Extraction-Free Multiplex Detection of Protein Biomarkers
Abstract
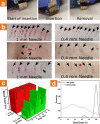
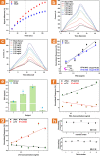
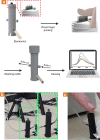
Protein biomarkers' detection is of utmost importance for preventive medicine and early detection of illnesses. Today, their detection relies entirely on clinical tests consisting of painful, invasive extraction of large volumes of venous blood; time-consuming postextraction sample manipulation procedures; and mostly label-based complex detection approaches. Here, we report on a point-of-care (POC) diagnosis paradigm based on the application of intradermal finger prick-based electronic nanosensors arrays for protein biomarkers' direct detection and quantification down to the sub-pM range, without the need for blood extraction and sample manipulation steps. The nanobioelectronic array performs biomarker sensing by a rapid intradermal prick-based sampling of proteins biomarkers directly from the capillary blood pool accumulating at the site of the microneedle puncture, requiring only 2 min and less than one microliter of a blood sample for a complete analysis. A 1 mm long microneedle element was optimal in allowing for pain-free dermal sampling with a 100% success rate of reaching and rupturing dermis capillaries. Current common micromachining processes and top-down fabrication techniques allow the nanobioelectronic sensor arrays to provide accurate and reliable clinical diagnostic results using multiple sensing elements in each microneedle and all-in-one direct and label-free multiplex biomarkers detection. Preliminary successful clinical studies performed on human volunteers demonstrated the ability of our intradermal, in-skin, blood extraction-free detection platform to accurately detect protein biomarkers as a plausible POC detection for future replacement of today's invasive clinical blood tests. This approach can be readily extended in the future to detect other clinically relevant circulating biomarkers, such as miRNAs, free-DNAs, exosomes, and small metabolites.
Keywords: Biomarkers; Biomolecules; Detection; Nanobioelectronics; Nanosensors.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



Similar articles
-
Transdermal Minimally Invasive Optical Multiplex Detection of Protein Biomarkers by Nanopillars Array-Embedded Microneedles.ACS Nano. 2024 Nov 5;18(44):30848-30862. doi: 10.1021/acsnano.4c11612. Epub 2024 Oct 28. ACS Nano. 2024. PMID: 39463189 Free PMC article.
-
Hydrogel-Coated Microneedle Arrays for Minimally Invasive Sampling and Sensing of Specific Circulating Nucleic Acids from Skin Interstitial Fluid.ACS Nano. 2019 Aug 27;13(8):9620-9628. doi: 10.1021/acsnano.9b04783. Epub 2019 Aug 16. ACS Nano. 2019. PMID: 31411871 Free PMC article.
-
Interstitial Fluid Biomarkers' Minimally Invasive Monitoring Using Microneedle Sensor Arrays.Anal Chem. 2022 Jan 18;94(2):968-974. doi: 10.1021/acs.analchem.1c03827. Epub 2021 Dec 22. Anal Chem. 2022. PMID: 34935347
-
Microneedle-based devices for point-of-care infectious disease diagnostics.Acta Pharm Sin B. 2021 Aug;11(8):2344-2361. doi: 10.1016/j.apsb.2021.02.010. Epub 2021 Feb 16. Acta Pharm Sin B. 2021. PMID: 34150486 Free PMC article. Review.
-
Circulating cell-free DNAs and miRNAs as promising non-invasive biomarkers for early detection of gastric cancer.Neoplasma. 2016;63(1):1-9. doi: 10.4149/neo_2016_002. Neoplasma. 2016. PMID: 26639229 Review.
Cited by
-
Implantable Electrochemical Microsensors for In Vivo Monitoring of Animal Physiological Information.Nanomicro Lett. 2023 Dec 12;16(1):49. doi: 10.1007/s40820-023-01274-4. Nanomicro Lett. 2023. PMID: 38087121 Free PMC article. Review.
-
Optimization of Surface Functionalizations for Ring Resonator-Based Biosensors.Sensors (Basel). 2024 May 14;24(10):3107. doi: 10.3390/s24103107. Sensors (Basel). 2024. PMID: 38793970 Free PMC article.
-
Transdermal Minimally Invasive Optical Multiplex Detection of Protein Biomarkers by Nanopillars Array-Embedded Microneedles.ACS Nano. 2024 Nov 5;18(44):30848-30862. doi: 10.1021/acsnano.4c11612. Epub 2024 Oct 28. ACS Nano. 2024. PMID: 39463189 Free PMC article.
-
Silk-based wearable devices for health monitoring and medical treatment.iScience. 2024 Mar 27;27(5):109604. doi: 10.1016/j.isci.2024.109604. eCollection 2024 May 17. iScience. 2024. PMID: 38628962 Free PMC article. Review.
-
Microneedle-Integrated Sensors for Extraction of Skin Interstitial Fluid and Metabolic Analysis.Int J Mol Sci. 2023 Jun 8;24(12):9882. doi: 10.3390/ijms24129882. Int J Mol Sci. 2023. PMID: 37373027 Free PMC article. Review.
References
-
- Jeun M.; Lee H. J.; Park S.; Do E.; Choi J.; Sung Y.-N.; Hong S.-M.; Kim S.-Y.; Kim D.-H.; Kang J. Y.; Son H.-N.; Joo J.; Song E. M.; Hwang S. W.; Park S. H.; Yang D.-H.; Ye B. D.; Byeon J.-S.; Choe J.; Yang S.-K.; et al. A Novel Blood-Based Colorectal Cancer Diagnostic Technology Using Electrical Detection of Colon Cancer Secreted Protein-2. Adv. Sci. 2019, 6, 1802115.10.1002/advs.201802115. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

