Anticoagulation in pediatric cancer-associated venous thromboembolism: a subgroup analysis of EINSTEIN-Jr
- PMID: 36006613
- PMCID: PMC9641171
- DOI: 10.1182/bloodadvances.2022008160
Anticoagulation in pediatric cancer-associated venous thromboembolism: a subgroup analysis of EINSTEIN-Jr
Abstract
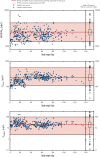
Anticoagulant treatment of pediatric cancer-associated venous thromboembolism (VTE) has not been prospectively evaluated. Management of anticoagulation for cancer-associated VTE is often challenged by drug interactions and treatment interruptions. A total of 56 of the 500 children (11.2%) with VTE who participated in the recent EINSTEIN-Jr randomized study had cancer (hematologic malignancy, 64.3%, solid malignant tumor, 35.7%). Children were allocated to either therapeutic-dose bodyweight-adjusted oral rivaroxaban (n=40) or standard anticoagulation with heparins, with or without vitamin K antagonists (n=16) and received a median of 30 concomitant medications. Based on sparse blood sampling at steady-state, pharmacokinetic (PK) parameters of rivaroxaban were derived using population PK modeling. During the 3 months of treatment, no recurrent VTE or major bleeding occurred (95% confidence interval, 0.0%-6.4%), and 3-month repeat imaging showed complete or partial vein recanalization in 20 and 24 of 52 evaluable children (38.5% and 46.2%, respectively). Anticoagulant treatment was interrupted 70 times in 26 (46.4%) children because of thrombocytopenia, invasive procedures, or adverse events, for a mean individual period of 5.8 days. Anticoagulant therapy was resumed in therapeutic doses and was not associated with thrombotic or bleeding complications. Rivaroxaban exposures were within the adult exposure range and similar to those observed in children with VTE who did not have cancer-associated VTE. Rivaroxaban and standard anticoagulants appeared safe and efficacious and were associated with reduced clot burden in most children with cancer-associated VTE, including those who had anticoagulant treatment interruptions. Rivaroxaban exposures were within the adult exposure range despite significant polypharmacy use. This trial was registered at www.clinicaltrials.gov as #NCT02234843.
© 2022 by The American Society of Hematology. Licensed under Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0), permitting only noncommercial, nonderivative use with attribution. All other rights reserved.
Conflict of interest statement
A.W.A.L., A.F.P., M.M., D.K., S.W., and K.T. are the employees of Bayer. C.M. reports personal fees and fees paid to his institution from Anthos, Bayer, Bristol-Myers Squibb, Janssen, Norgine, Pfizer, and Boehringer Ingelheim. H.v.O. reports fees paid to her institution from Bayer, Boehringer Ingelheim, and Octopharma. G.K. reports personal fees and fees paid to her institution from Bayer and Pfizer. The remaining authors declare no competing financial interests.
Figures
Similar articles
-
Safety and efficacy of anticoagulant therapy in pediatric catheter-related venous thrombosis (EINSTEIN-Jr CVC-VTE).Blood Adv. 2020 Oct 13;4(19):4632-4639. doi: 10.1182/bloodadvances.2020002637. Blood Adv. 2020. PMID: 33002131 Free PMC article.
-
Rivaroxaban for treatment of pediatric venous thromboembolism. An Einstein-Jr phase 3 dose-exposure-response evaluation.J Thromb Haemost. 2020 Jul;18(7):1672-1685. doi: 10.1111/jth.14813. Epub 2020 Jun 4. J Thromb Haemost. 2020. PMID: 32246743 Clinical Trial.
-
Bodyweight-adjusted rivaroxaban for children with venous thromboembolism (EINSTEIN-Jr): results from three multicentre, single-arm, phase 2 studies.Lancet Haematol. 2019 Oct;6(10):e500-e509. doi: 10.1016/S2352-3026(19)30161-9. Epub 2019 Aug 13. Lancet Haematol. 2019. PMID: 31420317 Clinical Trial.
-
Direct oral anticoagulants for extended treatment of venous thromboembolism: insights from the EINSTEIN CHOICE study.Blood Transfus. 2020 Jan;18(1):49-57. doi: 10.2450/2019.0265-18. Epub 2019 Apr 30. Blood Transfus. 2020. PMID: 31184579 Free PMC article. Review.
-
Venous Thromboembolism in Children: The Rivaroxaban Experience.Semin Thromb Hemost. 2024 Sep;50(6):866-872. doi: 10.1055/s-0043-1778106. Epub 2024 Jan 5. Semin Thromb Hemost. 2024. PMID: 38181816 Review.
Cited by
-
Efficacy and safety of antithrombotic therapy for preventing and treating pediatric thromboembolic disease: a systematic review.Sci Rep. 2024 Jun 11;14(1):13378. doi: 10.1038/s41598-024-64334-8. Sci Rep. 2024. PMID: 38862574 Free PMC article.
-
The value of the old and the new.Blood Adv. 2023 Jul 25;7(14):3683-3685. doi: 10.1182/bloodadvances.2023009723. Blood Adv. 2023. PMID: 36939386 Free PMC article. No abstract available.
-
Fungal Pneumonia and Pulmonary Embolism in a Child with Refractory Leukemia: Coincidence or Not?Indian J Hematol Blood Transfus. 2025 Apr;41(2):443-446. doi: 10.1007/s12288-024-01823-w. Epub 2024 Jul 20. Indian J Hematol Blood Transfus. 2025. PMID: 40224686 No abstract available.
-
A global survey on practice changes and barriers to the use of direct oral anticoagulants in children.Blood Adv. 2025 Aug 12;9(15):3718-3727. doi: 10.1182/bloodadvances.2025016681. Blood Adv. 2025. PMID: 40353824 Free PMC article.
-
Systematic Review of Randomized Clinical Trials on Direct Oral Anticoagulants in Pediatric Heart Diseases.Clin Appl Thromb Hemost. 2024 Jan-Dec;30:10760296241271974. doi: 10.1177/10760296241271974. Clin Appl Thromb Hemost. 2024. PMID: 39099474 Free PMC article.
References
-
- Prandoni P., Lensing A.W., Piccioli A., et al. Recurrent venous thromboembolism and bleeding complications during anticoagulant treatment in patients with cancer and venous thrombosis. Blood. 2002;100:3484–3488. - PubMed
-
- Prins M.H., Lensing A.W., Brighton T.A., et al. Oral rivaroxaban versus enoxaparin with vitamin K antagonist for the treatment of symptomatic venous thromboembolism in patients with cancer (EINSTEIN-DVT and EINSTEIN-PE): a pooled subgroup analysis of two randomised controlled trials. Lancet Haematol. 2014;1:E37–E46. - PubMed
-
- Shaw J.R., Douketis J., Le Gal G., Carrier M. Periprocedural interruption of anticoagulation in patients with cancer-associated venous thromboembolism: an analysis of thrombotic and bleeding complications. J Thromb Haemostasis. 2019;17:1171–1178. - PubMed
-
- Male C., Lensing A.W.A., Palumbo J., et al. Randomised controlled trial of rivaroxaban compared to standard anticoagulants for the treatment of acute venous thromboembolism in children. Lancet Haematology. 2020;7:e18–e27. - PubMed
-
- Monagle P., Lensing A.W.A., Thelen K., et al. Bodyweight-adjusted rivaroxaban in children with venous thromboembolism. An Einstein-Jr. phase II evaluation. Lancet Haematol. 2019;6:e500–e509. - PubMed

