Click Step-Growth Polymerization and E/ Z Stereochemistry Using Nucleophilic Thiol-yne/-ene Reactions: Applying Old Concepts for Practical Sustainable (Bio)Materials
- PMID: 36006902
- PMCID: PMC9454099
- DOI: 10.1021/acs.accounts.2c00293
Click Step-Growth Polymerization and E/ Z Stereochemistry Using Nucleophilic Thiol-yne/-ene Reactions: Applying Old Concepts for Practical Sustainable (Bio)Materials
Abstract
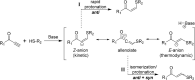
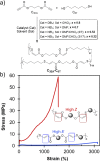
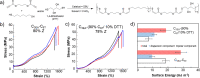
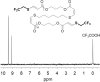
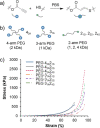
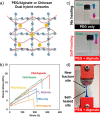
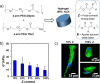
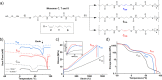
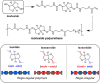
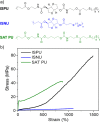
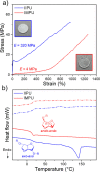
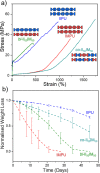
Polymer sustainability is synonymous with "bioderived polymers" and the zeitgeist of "using renewable feedstocks". However, this sentiment does not adequately encompass the requirements of sustainability in polymers. In addition to recycling considerations and mechanical performance, following green chemistry principles also needs to be maximized to improve the sustainability of polymer synthesis. The synthetic cost (i.e., maximizing atom economy, reducing chemical hazards, and lowering energy requirements) of producing polymers should be viewed as equally important to the monomer source (biomass vs petrol platform chemicals). Therefore, combining the use of renewable feedstocks with efficient syntheses and green chemistry principles is imperative to delivering truly sustainable polymers. The high efficiency, atom economy, and single reaction trajectories that define click chemistry reactions position them as ideal chemical approaches to synthesize polymers in a sustainable manner while simultaneously expanding the structural scope of accessible polymers from sustainably sourced chemicals.Click step-growth polymerization using the thiol-yne Michael addition, a reaction first reported over a century ago, has emerged as an extremely mild and atom-efficient pathway to yield high-performance polymers with controllable E/Z stereochemistry along the polymer backbone. Building on studies of aromatic thiol-yne polymers, around 10 years ago our group began investigating the thiol-yne reaction for the stereocontrolled synthesis of alkene-containing aliphatic polyesters. Our early studies established a convenient path to high-molecular-weight (>100 kDa) E-rich or Z-rich step-growth polymers by judiciously changing the catalyst and/or reaction solvent. This method has since been adapted to synthesize fast-degrading polyesters, high-performance polyamides, and resilient hydrogel biomaterials. Across several systems, we have observed dramatic differences in material properties among polymers with different alkene stereochemistry.We have also explored the analogous thiol-ene Michael reaction to create high-performance poly(ester-urethanes) with precise E/Z stereochemistry. In contrast to the stereoselective thiol-yne polymerization, here the use of monomers with predefined E/Z (geometric) isomerism (arising from either alkenes or the planar rigidity of ring units) affords polymers with total control over stereochemistry. This advancement has enabled the synthesis of tough, degradable materials that are derived from sustainable monomer feedstocks. Employing isomers of sugar-derived isohexides, bicyclic rigid-rings possessing geometric isomerism, led to degradable polymers with fundamentally opposing mechanical behavior (i.e., plastic vs elastic) simply by adjusting the stereochemistry of the isohexide.In this Account, we feature our investigation of thiol-yne/-ene click step-growth polymers and efforts to establish structure-property relationships toward degradable materials with practical mechanical performance in the context of sustainable polymers and/or biomaterials. We have paid attention to installing and controlling geometric isomerism by using these click reactions, an overarching objective of our work in this research area. The exquisite control of geometric isomerism that is possible within polymer backbones, as enabled by convenient click chemistry reactions, showcases a powerful approach to creating multipurpose degradable polymers.
Conflict of interest statement
The authors declare the following competing financial interest(s): A.P.D. is named as an inventor of a patent application based on the thiol-yne nucleophilic addition work; both J.C.W. and A.P.D. are named as inventors of a provisional patent application based on the isohexide polymer work.
Figures



References
-
- Worch J. C.; Weems A. C.; Yu J.; Arno M. C.; Wilks T. R.; Huckstepp R. T. R.; O’Reilly R. K.; Becker M. L.; Dove A. P. Elastomeric polyamide biomaterials with stereochemically tuneable mechanical properties and shape memory. Nat. Commun. 2020, 11, 3250. 10.1038/s41467-020-16945-8. - DOI - PMC - PubMed
-
- Geng Z.; Shin J. J.; Xi Y.; Hawker C. J. Click chemistry strategies for the accelerated synthesis of functional macromolecules. J. Polym. Sci. 2021, 59, 963–1042. 10.1002/pol.20210126. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

