Safety and performance of surgical adhesives
- PMID: 36006958
- PMCID: PMC9409600
- DOI: 10.1371/journal.pone.0271531
Safety and performance of surgical adhesives
Erratum in
-
Correction: Safety and performance of surgical adhesives.PLoS One. 2023 Nov 15;18(11):e0294614. doi: 10.1371/journal.pone.0294614. eCollection 2023. PLoS One. 2023. PMID: 37967064 Free PMC article.
Abstract
Background: Tissue adhesives are an alternative to conventional surgical sutures to reduce the time and cost of wound closure and to improve patient comfort. The use of tissue adhesives does not require any subsequent intervention and significantly lowers the volume and rate of blood loss, and reduces the need for transfusions during and after surgery. However, based on their formulation, tissue adhesives' safety profile and functional properties may differ. Therefore, this study aimed to evaluate the basic safety and performance of NE'X Glue® Surgical Sealant, BioGlue® Surgical Sealant, and PREVELEAKTM Surgical Sealant in vitro.
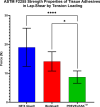
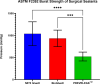
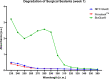
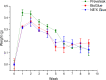
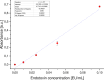
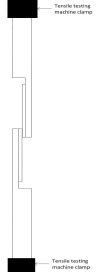
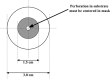
Methods: The basic safety of commercially available tissue adhesives was evaluated using MEM elution assay according to ISO 10993-5 and endotoxin level according to 85. USP. The in vitro performance was evaluated using lap-shear by tension loading test, burst strength test, degradation, and swelling assays.
Results: NE'X Glue®, BioGlue®, and PREVELEAKTM did not cause cytotoxicity in MEM elution assay. All surgical adhesives are below the general limit of endotoxin contamination of 20 EU/device. NE'X Glue® and BioGlue® showed the highest and comparable strength properties in lap shear and burst strength tests compared to PREVELEAKTM. NE'X Glue® and PREVELEAKTM are characterized by lower degradation potential than BioGlue®. PREVELEAKTM is characterized by the highest swelling when compared to NE'X Glue® and BioGlue®.
Conclusions: NE'X Glue® is most versatile in terms of functional properties while maintaining the same safety profile as BioGlue® and PREVELEAKTM.
Conflict of interest statement
Dr. Damian Matak and Dr. Lukasz Szymanski have invented NE’X Glue®. All tests were per-formed in ISO 17025 accredited and GLP certified laboratory to ensure data integrity. Dr. Szymanski and Dr. Matak have no financial interests in NE’X Glue® Surgical Adhesive. This does not alter our adherence to PLOS ONE policies on sharing data and materials.
Figures


Similar articles
-
Biocompatibility of novel albumin-aldehyde surgical adhesive.Sci Rep. 2022 Jul 26;12(1):12749. doi: 10.1038/s41598-022-16853-5. Sci Rep. 2022. PMID: 35882896 Free PMC article.
-
Investigation of surgical adhesives for vocal fold wound closure.Laryngoscope. 2019 Sep;129(9):2139-2146. doi: 10.1002/lary.27769. Epub 2019 Mar 18. Laryngoscope. 2019. PMID: 30883775 Free PMC article.
-
Histological effects of fibrin glue and synthetic tissue glues on the spinal cord: are they safe to use?Br J Neurosurg. 2017 Dec;31(6):695-700. doi: 10.1080/02688697.2017.1359491. Epub 2017 Aug 28. Br J Neurosurg. 2017. PMID: 28847164
-
Subcuticular sutures for skin closure in non-obstetric surgery.Cochrane Database Syst Rev. 2020 Apr 9;4(4):CD012124. doi: 10.1002/14651858.CD012124.pub2. Cochrane Database Syst Rev. 2020. PMID: 32271475 Free PMC article.
-
Tissue Adhesives for the Management of Corneal Perforations and Challenging Corneal Conditions.Clin Ophthalmol. 2023 Jan 15;17:209-223. doi: 10.2147/OPTH.S394454. eCollection 2023. Clin Ophthalmol. 2023. PMID: 36685088 Free PMC article. Review.
Cited by
-
Correction: Safety and performance of surgical adhesives.PLoS One. 2023 Nov 15;18(11):e0294614. doi: 10.1371/journal.pone.0294614. eCollection 2023. PLoS One. 2023. PMID: 37967064 Free PMC article.
References
-
- Swathi Krishna KV, Uma Maheswari LS, Rajeswari G. Surgical glue- a promising technology for wound healing. Int J Res Pharm Sci Technol. 2018;1: 9–11. doi: 10.33974/ijrpst.v1i1.18 - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

