The incidence of severe oral mucositis in patients undergoing different conditioning regimens in haematopoietic stem cell transplantation
- PMID: 36008731
- PMCID: PMC9410738
- DOI: 10.1007/s00520-022-07328-4
The incidence of severe oral mucositis in patients undergoing different conditioning regimens in haematopoietic stem cell transplantation
Abstract
Purpose: Oral mucositis is a common complication during haematopoietic stem cell transplantation (HSCT). This study aimed to assess the incidence of severe mucositis in patients undergoing different HSCT regimens.
Methods: This single-centre retrospective study reviewed daily oral assessment for 467 consecutive patients who underwent different transplant regimens for matched unrelated or related allogeneic HSCT with post-transplant methotrexate, haploidentical or mismatched HSCT with post-transplant cyclophosphamide (PTCy), or autologous HSCT. Oral care and cryotherapy with melphalan were used. Patient demographic data, oral mucositis WHO grade, use of total parenteral nutrition (TPN) and patient-controlled analgesia (PCA) were collected.
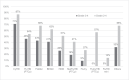
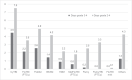
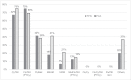
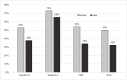
Results: Grade 3-4 oral mucositis was common in myeloablative total body irradiation (TBI)-based regimens cyclophosphamide/ TBI (CyTBI) (71%) and fludarabine/ TBI (FluTBI) with PTCy (46%), as well as reduced-intensity fludarabine/melphalan (FluMel) (43%) and carmustine/etoposide/cytarabine/melphalan (BEAM) autologous HSCT (41%). In contrast, grade 3-4 oral mucositis was less common in reduced-intensity haploidentical regimen melphalan/fludarabine/TBI with PTCy (19%), all non-myeloablative regimens (0-9%) and high-dose melphalan autologous HSCT (26%). TPN and PCA use were correlated to oral mucositis severity.
Conclusions: Severe oral mucositis was associated with myeloablative TBI, methotrexate and melphalan in combination with methotrexate and in BEAM. Use of PTCy was preferable over methotrexate to prevent oral mucositis.
Keywords: Conditioning; Haematopoietic stem cell transplantation; Oral mucositis; Risk factors.
© 2022. Crown.
Conflict of interest statement
This study was conducted as a part of principal author’s Doctor of Philosophy degree. She has received a scholarship from the Royal Brisbane and Women’s Hospital Foundation. Other authors declare no competing financial interests.
Figures

References
-
- Bellm LA, Epstein JB, Rose-Ped A, Martin P, Fuchs HJ. Patient reports of complications of bone marrow transplantation. Support Care Cancer. 2000;8(1):33–39. - PubMed
-
- Vokurka S, Bystrická E, Koza V, Scudlová J, Pavlicová V, Valentová D, Visokaiová M, Misaniová L. Higher incidence of chemotherapy induced oral mucositis in females: a supplement of multivariate analysis to a randomized multicentre study. Support Care Cancer. 2006;14(9):974–976. doi: 10.1007/s00520-006-0031-z. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

