Effect of Human Platelet Lysate as Cultivation Nutrient Supplement on Human Natal Dental Pulp Stem Cell In Vitro Expansion
- PMID: 36008985
- PMCID: PMC9405745
- DOI: 10.3390/biom12081091
Effect of Human Platelet Lysate as Cultivation Nutrient Supplement on Human Natal Dental Pulp Stem Cell In Vitro Expansion
Abstract
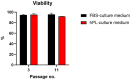
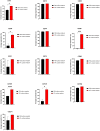
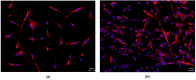
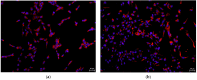
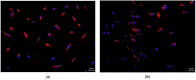
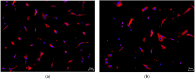
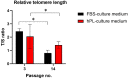
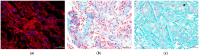
Despite several scientific or ethical issues, fetal bovine serum (FBS) remains the standard nutrient supplement in the mesenchymal stem cell cultivation medium. Cell amplification plays an important role in human stem cell therapies. Increasing interest in this field has supported attempts to find suitable human alternatives to FBS for in vitro cell propagation. Human platelet lysate (hPL) has recently been determined as one of them. Our study aimed to evaluate the influence of 2% hPL in the growth medium for in vitro expansion of human natal dental pulp stem cells (hNDP-SCs). The effect was determined on proliferation rate, viability, phenotype profile, expression of several markers, relative telomere length change, and differentiation potential of four lineages of hNDP-SCs. As a control, hNDP-SCs were simultaneously cultivated in 2% FBS. hNDP-SCs cultivated in hPL showed a statistically significantly higher proliferation rate in initial passages. We did not observe a statistically significant effect on mesenchymal stem cell marker (CD29, CD44, CD73, CD90) or stromal-associated marker (CD13, CD166) expression. The cell viability, relative telomere length, or multipotency remained unaffected in hNDP-SCs cultivated in hPL-medium. In conclusion, hPL produced under controlled and standardized conditions is an efficient serum supplement for in vitro expansion of hNDP-SCs.
Keywords: culture medium nutrient supplement; fetal bovine serum; human natal stem cells; human platelet lysate; mesenchymal stem cells; regenerative medicine; stem cell cultivation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

