Advancing Our Understanding of the Chronically Denervated Schwann Cell: A Potential Therapeutic Target?
- PMID: 36009023
- PMCID: PMC9406133
- DOI: 10.3390/biom12081128
Advancing Our Understanding of the Chronically Denervated Schwann Cell: A Potential Therapeutic Target?
Abstract
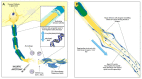
Outcomes for patients following major peripheral nerve injury are extremely poor. Despite advanced microsurgical techniques, the recovery of function is limited by an inherently slow rate of axonal regeneration. In particular, a time-dependent deterioration in the ability of the distal stump to support axonal growth is a major determinant to the failure of reinnervation. Schwann cells (SC) are crucial in the orchestration of nerve regeneration; their plasticity permits the adoption of a repair phenotype following nerve injury. The repair SC modulates the initial immune response, directs myelin clearance, provides neurotrophic support and remodels the distal nerve. These functions are critical for regeneration; yet the repair phenotype is unstable in the setting of chronic denervation. This phenotypic instability accounts for the deteriorating regenerative support offered by the distal nerve stump. Over the past 10 years, our understanding of the cellular machinery behind this repair phenotype, in particular the role of c-Jun, has increased exponentially, creating opportunities for therapeutic intervention. This review will cover the activation of the repair phenotype in SC, the effects of chronic denervation on SC and current strategies to 'hack' these cellular pathways toward supporting more prolonged periods of neural regeneration.
Keywords: Schwann cells; c-Jun; chronic Schwann cell denervation; nerve injury; peripheral nerve injury; peripheral nerve regeneration.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Miller C., Peek A.L., Power D., Heneghan N.R. Psychological consequences of traumatic upper limb peripheral nerve injury: A systematic review. Hand Ther. 2017;22:35–45. doi: 10.1177/1758998316679387. - DOI
-
- Aegineta P. The seven books of Paulus Aegineta. Volume 4 Sydenham Society; Sydenham, UK: 1844.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

