Evaluation of the Anti- Leishmania mexicana and - Trypanosoma brucei Activity and Mode of Action of 4,4'-(Arylmethylene)bis(3-methyl-1-phenyl-1 H-pyrazol-5-ol)
- PMID: 36009460
- PMCID: PMC9405596
- DOI: 10.3390/biomedicines10081913
Evaluation of the Anti- Leishmania mexicana and - Trypanosoma brucei Activity and Mode of Action of 4,4'-(Arylmethylene)bis(3-methyl-1-phenyl-1 H-pyrazol-5-ol)
Abstract
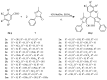
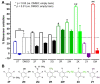
Trypanosomiasis and leishmaniasis are neglected infections caused by trypanosomatid parasites. The first-line treatments have many adverse effects, high costs, and are prone to resistance development, hence the necessity for new chemotherapeutic options. In line with this, twenty five 4,4′-(arylmethylene)bis(1H-pyrazol-5-ols) derivatives were synthesized and evaluated in vitro for their anti-trypanosomatid activity. Ten and five compounds from this series showed IC50 ≤ 10 µM against the promastigote and the bloodstream stage of Leishmania mexicana and Trypanosoma brucei brucei, respectively. Overall, derivatives with pyrazole rings substituted with electron-withdrawing groups proved more active than those with electron-donating groups. The hits proved moderately selective towards L. mexicana and T. brucei (selectivity index, SI, compared to murine macrophages = 5−26). The exception was one derivative displaying an SI (>111−189) against T. brucei that surpassed, by >6-fold, the selectivity of the clinical drug nifurtimox (SI = 13−28.5). Despite sharing a common scaffold, the hits differed in their mechanism of action, with halogenated derivatives inducing a rapid and marked intracellular oxidative milieu in infective T. brucei. Notably, most of the hits presented better absorption, distribution, metabolism, and excretion (ADME) properties than the reference drugs. Several of the bioactive molecules herein identified represent a promising starting point for further improvement of their trypanosomatid potency and selectivity.
Keywords: 4,4′-(arylmethylene)bis(1H-pyrazol-5-ols); ADME; Leishmania; Trypanosoma; drug-like; redox biosensor.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures

Similar articles
-
New multifunctional Ru(II) organometallic compounds show activity against Trypanosoma brucei and Leishmania infantum.J Inorg Biochem. 2022 Dec;237:112016. doi: 10.1016/j.jinorgbio.2022.112016. Epub 2022 Oct 3. J Inorg Biochem. 2022. PMID: 36244312
-
New heterobimetallic ferrocenyl derivatives: Evaluation of their potential as prospective agents against trypanosomatid parasites and Mycobacterium tuberculosis.J Inorg Biochem. 2018 Oct;187:73-84. doi: 10.1016/j.jinorgbio.2018.07.013. Epub 2018 Jul 23. J Inorg Biochem. 2018. PMID: 30055398
-
Synthesis of 4,4'-(arylmethylene)bis(3-methyl-1-phenyl-1H-pyrazol-5-ols) and evaluation of their antioxidant and anticancer activities.BMC Chem. 2021 Jun 3;15(1):38. doi: 10.1186/s13065-021-00765-y. BMC Chem. 2021. PMID: 34082794 Free PMC article.
-
Naphthoquinones and Derivatives for Chemotherapy: Perspectives and Limitations of their Anti-trypanosomatids Activities.Curr Pharm Des. 2021;27(15):1807-1824. doi: 10.2174/1381612826666201109111802. Curr Pharm Des. 2021. PMID: 33167829 Review.
-
The use of Sulfonamide Derivatives in the Treatment of Trypanosomatid Parasites including Trypanosoma cruzi, Trypanosoma brucei, and Leishmania ssp.Med Chem. 2020;16(1):24-38. doi: 10.2174/1573406415666190620141109. Med Chem. 2020. PMID: 31218962 Review.
Cited by
-
A Promising Amphotericin B Derivative Induces Morphological Alterations, Mitochondrial Damage, and Oxidative Stress In Vitro and Prevents Mice from Death Produced by a Virulent Strain of Trypanosoma cruzi.Microorganisms. 2024 May 24;12(6):1064. doi: 10.3390/microorganisms12061064. Microorganisms. 2024. PMID: 38930447 Free PMC article.
References
-
- World Health Organization . WHO Technical Report Series. Volume 975. WHO Press; Geneve, Switzerland: 2012. Research priorities for Chagas disease, Human African Trypanosomiasis and Leishmaniasis: Technical report of the TDR Disease Reference Group on Chagas Disease, Human African Trypanosomiasis and Leishmaniasis; pp. 26–29. - PubMed
-
- World Health Organization . WHO Technical Report Series. Volume 949. WHO Press; Geneve, Switzerland: 2010. World Health Organization Control of the Leishmaniasis: Report of a meeting of the WHO Expert Committee on the control of Leishmaniases, Geneva, 22–26 March 2010; pp. 54–74.
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

