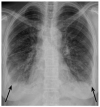
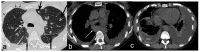
Figure 2
Axial non-contrast chest CT showed (a) multiple bilateral lung nodules (big arrows) and foci of thickened interlobular septa (small arrows); (b) enlarged subcarinal lymph nodes (white arrow); (c) right-sided pleural effusion with pleural thickening (measured) and left-sided pleural effusion partially compressing underlying lung parenchyma. Enlarged left supraclavicular and hepatic lymph nodes were found in the ultrasound examination. The overall clinical and radiologic appearance was suggestive of a lymphoma. On the day of admission, the patient developed fever of 40 °C. Sputum, blood, and urine cultures were taken—all turned out to be negative. Neither Legionella antigen in urine sample nor CMV antigen in blood were detected. An HIV test was requested as a part of the differential diagnosis of lymphopenia and lymphadenopathy. Meanwhile, an empirical antibiotic therapy consisting of ceftriaxone and levofloxacin was commenced; it was soon extended by the inclusion of clindamycin because the patient continued to be febrile. Bronchoscopy was performed and it showed a scar from previous lymph node ulceration into the left upper bronchus; aside from this the macroscopic appearance of the airways was irrelevant. The fluid from bronchoalveolar lavage (BALF) was collected and cultured. Direct microscopic examination was negative for acid-fast bacilli, but molecular testing with GeneXpert MTB/RIF assay detected the presence of Mycobacterium tuberculosis complex DNA; no rifampicin resistance gene was found. At the same time, a positive result for HIV-1 test (Bio-Rad Geenius TM HIV 1/2 Confirmatory Assay) was obtained. Anti-tuberculous treatment was started with isoniazid, rifampicin, ethambutol, and pyrazinamide. Soon, the patient became apyrexial and CRP started to decrease gradually. Mycobacterium tuberculosis (MTB) strain, susceptible to all primary anti-TB drugs, was cultured from BALF after two weeks. At the time of HIV infection diagnosis, the CD4+ T-lymphocyte count was 107 cells/mm3, and viral load 13,846 copies/mm3. Antiretroviral therapy (ART) with emtricitabine, tenofovir, and dolutegravir was commenced 11 days after anti-TB treatment was started. It resulted in a gradual improvement in CD4+ cell count and viral suppression; after four months CD4+ T-lymphocyte count reached level of >400 cells/mm3. Both anti-TB and antiviral therapies were well tolerated. The four-drug anti-TB treatment was continued for two months and was followed by seven months of isoniazid and rifampicin. Follow-up chest HRCT showed the regression of mediastinal lymphadenopathy and significant improvement of parenchymal and pleural abnormalities. The series of sputum cultures for acid-fast bacilli was negative. Three years after the diagnosis of pulmonary TB and acquired immunodeficiency syndrome (AIDS), the patient remains in good condition and continues ART successfully in an HIV/AIDS-dedicated out-patient clinic. The reported patient with newly diagnosed HIV infection and pulmonary TB had no background chronic conditions and considered herself healthy until the unspecific chest and feet pains, mild dry cough, fatigue, and reduced appetite occurred. The symptoms of pulmonary TB are unspecific, and the most common include cough with scanty sputum, haemoptysis, dyspnoea, chest pain, low-grade intermittent fever, sweating, fatigue, and weight loss [10]. Pulmonary TB in HIV-positive people presents with similar symptoms, but lower CD4+ cell counts are associated with more severe systemic symptoms [9,11]. Typical chest X-ray findings in pulmonary TB are: upper lung zones patchy consolidations, cavitations, features of bronchial dissemination [12]. Such radiologic features were not found in the presented patient. Chest CT scan showed multiple small nodules with perilymphatic distribution, interlobular septa thickening, enlarged mediastinal lymph nodes, and bilateral pleural effusion. As the HIV status of the patient was not known at that time, the radiologists suggested lung involvement in the course of a lymphoproliferative disease. That impression was further augmented by the presence of abdominal and supraclavicular lymphadenopathy. Nonspecific symptoms, high inflammatory indices, the presence of lymphopenia, lymphadenopathy, and pulmonary and pleural lesions required broad diagnostic work-up. Testing for HIV and MTB gave the conclusive outcome, and pulmonary TB in the course of HIV infection was diagnosed. Radiologic phenotype of TB associated with HIV is similar to that without HIV co-infection when CD4+ cell count is high, i.e., >350 cells/mm3. In cases with lower CD4+ cell counts, the presentation of pulmonary TB is shifted towards atypical patterns, such as parenchymal consolidations affecting the middle and lower lung zones, miliary infiltrates, chest lymphadenopathy, and pleural effusion, as in the presented patient [6,7,8,9]. Lung disease in the late period of HIV infection may be caused by opportunistic bacteria, tuberculous and non-tuberculous mycobacteria, viral and fungal pathogens, but also by the spectrum of neoplastic disorders, such as non-Hodgkin’s lymphoma or Kaposi’s sarcoma [13]. They may present with pleural effusion and pulmonary nodular infiltrates [13,14]. Thus, the differential considerations in the reported patient should also include these neoplastic diseases. The bronchoscopy revealed no suspicious endobronchial lesions, and microscopic examination of BALF was negative for atypical or neoplastic cells. Finally, therapies targeted at TB and HIV resulted in resolution of lymphadenopathy. An additional difficulty in diagnosing TB in PLWH is a higher rate of smear-negative disease. For this reason, molecular WHO-recommended rapid diagnostic tests, such as GeneXpert MTB/RIF assay, are recommended as an initial test rather than smear microscopy or culture [3]. Anti-tuberculous treatment regimen and its duration in drug-susceptible TB in PLWH are the same as in TB without HIV co-infection [5]. ART should be started as soon as possible (within two weeks of initiating TB treatment) regardless of CD4+ cell count, with the only exception being TB meningitis, where it should be delayed for four weeks if CD4+ < 50 (100) cells/mm3 [5]. The risk of death in people co-infected with HIV and TB is reportedly two to five times higher than in HIV-infected patients without TB with matched CD4+ cell counts, irrespective of ART, effective TB treatment, and good access to healthcare [15,16,17]. There is the evidence that TB directly contributes to mortality in HIV-infected patients, rather than simply presenting as a marker of advanced immunodeficiency [18]. Delayed TB diagnosis, i.e., after ≥1 month of symptoms duration, increases the risk of death [19], and the first 3 months after TB diagnosis seem crucial for survival in PLWH [15]. A TB recurrence rate is higher in PLWH than in people without HIV—4.5 vs. 1.9 per 100 person-years, respectively [20]. The presented patient remains well three years after diagnosis of TB and HIV co-infection, with viral suppression on ART and no signs of TB recurrence. Populations with high prevalence of HIV infection include men who have sex with men, intravenous drug users, people in prisons and other closed settings, sex workers, and transgender people [21]. In summary, co-infection with HIV may change the clinical phenotype of TB, leading to diagnostic problems and delayed treatment. The highest level of vigilance with regard to TB is recommended in PLWH. Moreover, each newly diagnosed TB patient should be tested for HIV.