Si-Wu Water Extracts Protect against Colonic Mucus Barrier Damage by Regulating Muc2 Mucin Expression in Mice Fed a High-Fat Diet
- PMID: 36010498
- PMCID: PMC9407452
- DOI: 10.3390/foods11162499
Si-Wu Water Extracts Protect against Colonic Mucus Barrier Damage by Regulating Muc2 Mucin Expression in Mice Fed a High-Fat Diet
Abstract
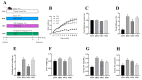
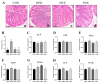
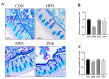
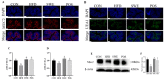
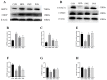
A high-fat diet (HFD) could cause gut barrier damage. The herbs in si-wu (SW) include dang gui (Angelica sinensis (Oliv.) Diels), shu di huang (the processed root of Rehmannia glutinosa Libosch.), chuan xiong (rhizome of Ligusticum chuanxiong Hort.), and bai shao (the root of Paeonia lactiflora f. pilosella (Nakai) Kitag.). Si-wu water extracts (SWE) have been used to treat blood deficiency. Components of one herb from SW have been reported to have anti-inflammatory and anti-obesity activities. However, there have been no reports about the effects of SWE on gut barrier damage. Therefore, the aim of the study was to explore the effect of SWE on gut barrier damage. In this study, we found that SWE effectively controlled body weight, liver weight, and feed efficiency, as well as decreased the serum TC level in HFD-fed mice. Moreover, SWE and rosiglitazone (Ros, positive control) increased the colonic alkaline phosphatase (ALP) level, down-regulated serum pro-inflammatory cytokine levels, and reduced intestinal permeability. In addition, SWE increased goblet cell numbers and mucus layer thickness to strengthen the mucus barrier. After supplementation with SWE and rosiglitazone, the protein expression of CHOP and GRP78 displayed a decrease, which improved the endoplasmic reticulum (ER) stress condition. Meanwhile, the increase in Cosmc and C1GALT1 improved the O-glycosylation process for correct protein folding. These results collectively demonstrated that SWE improved the mucus barrier, focusing on Muc2 mucin expression, in a prolonged high-fat diet, and provides evidence for the potential of SWE in the treatment of intestinal disease-associated mucus barrier damage.
Keywords: O-glycosylation; endoplasmic reticulum stress; high-fat diet; mucus barrier; si-wu water extract.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Citrus peel ameliorates mucus barrier damage in HFD-fed mice.J Nutr Biochem. 2023 Feb;112:109206. doi: 10.1016/j.jnutbio.2022.109206. Epub 2022 Nov 9. J Nutr Biochem. 2023. PMID: 36370925
-
IL-10 promotes production of intestinal mucus by suppressing protein misfolding and endoplasmic reticulum stress in goblet cells.Gastroenterology. 2013 Feb;144(2):357-368.e9. doi: 10.1053/j.gastro.2012.10.043. Epub 2012 Oct 31. Gastroenterology. 2013. PMID: 23123183
-
Stress disrupts intestinal mucus barrier in rats via mucin O-glycosylation shift: prevention by a probiotic treatment.Am J Physiol Gastrointest Liver Physiol. 2014 Aug 15;307(4):G420-9. doi: 10.1152/ajpgi.00290.2013. Epub 2014 Jun 26. Am J Physiol Gastrointest Liver Physiol. 2014. PMID: 24970779
-
The current evidence for the treatment of sepsis with Xuebijing injection: Bioactive constituents, findings of clinical studies and potential mechanisms.J Ethnopharmacol. 2021 Jan 30;265:113301. doi: 10.1016/j.jep.2020.113301. Epub 2020 Aug 27. J Ethnopharmacol. 2021. PMID: 32860891 Review.
-
Impact of Western Diet and Ultra-Processed Food on the Intestinal Mucus Barrier.Biomedicines. 2023 Jul 18;11(7):2015. doi: 10.3390/biomedicines11072015. Biomedicines. 2023. PMID: 37509654 Free PMC article. Review.
Cited by
-
Clinical glycoproteomics: methods and diseases.MedComm (2020). 2024 Oct 4;5(10):e760. doi: 10.1002/mco2.760. eCollection 2024 Oct. MedComm (2020). 2024. PMID: 39372389 Free PMC article. Review.
-
Characteristics of mucin hypersecretion in different inflammatory patterns based on endotypes of chronic rhinosinusitis.Clin Transl Allergy. 2024 Jan;14(1):e12334. doi: 10.1002/clt2.12334. Clin Transl Allergy. 2024. PMID: 38282195 Free PMC article. Review.
-
Capsazepine-Induced Altered Colonic Mucosal Health Limits Isomalto-oligosaccharide Action in High-Fat Diet-Fed C57BL/6J Mice.ACS Pharmacol Transl Sci. 2023 Apr 3;6(4):600-613. doi: 10.1021/acsptsci.2c00243. eCollection 2023 Apr 14. ACS Pharmacol Transl Sci. 2023. PMID: 37082749 Free PMC article.
References
-
- Lassenius M.I., Pietiläinen K.H., Kaartinen K., Pussinen P.J., Syrjänen J., Forsblom C., Pörsti I., Rissanen A., Kaprio J., Mustonen J., et al. Bacterial endotoxin activity in human serum is associated with dyslipidemia, insulin resistance, obesity, and chronic inflammation. Diabetes Care. 2011;34:1809–1815. doi: 10.2337/dc10-2197. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

