Characterization of Calpain and Caspase-6-Generated Glial Fibrillary Acidic Protein Breakdown Products Following Traumatic Brain Injury and Astroglial Cell Injury
- PMID: 36012232
- PMCID: PMC9409281
- DOI: 10.3390/ijms23168960
Characterization of Calpain and Caspase-6-Generated Glial Fibrillary Acidic Protein Breakdown Products Following Traumatic Brain Injury and Astroglial Cell Injury
Abstract
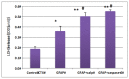
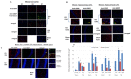
Glial fibrillary acidic protein (GFAP) is the major intermediate filament III protein of astroglia cells which is upregulated in traumatic brain injury (TBI). Here we reported that GFAP is truncated at both the C- and N-terminals by cytosolic protease calpain to GFAP breakdown products (GBDP) of 46-40K then 38K following pro-necrotic (A23187) and pro-apoptotic (staurosporine) challenges to primary cultured astroglia or neuron-glia mixed cells. In addition, with another pro-apoptotic challenge (EDTA) where caspases are activated but not calpain, GFAP was fragmented internally, generating a C-terminal GBDP of 20 kDa. Following controlled cortical impact in mice, GBDP of 46-40K and 38K were formed from day 3 to 28 post-injury. Purified GFAP protein treated with calpain-1 and -2 generates (i) major N-terminal cleavage sites at A-56*A-61 and (ii) major C-terminal cleavage sites at T-383*Q-388, producing a limit fragment of 38K. Caspase-6 treated GFAP was cleaved at D-78/R-79 and D-225/A-226, where GFAP was relatively resistant to caspase-3. We also derived a GBDP-38K N-terminal-specific antibody which only labels injured astroglia cell body in both cultured astroglia and mouse cortex and hippocampus after TBI. As a clinical translation, we observed that CSF samples collected from severe human TBI have elevated levels of GBDP-38K as well as two C-terminally released GFAP peptides (DGEVIKES and DGEVIKE). Thus, in addition to intact GFAP, both the GBDP-38K as well as unique GFAP released C-terminal proteolytic peptides species might have the potential in tracking brain injury progression.
Keywords: GFAP; astroglial injury; biomarkers; calpain; caspase; traumatic brain injury.
Conflict of interest statement
K.K.W. and W.E.H. are share-holder of Gryphon Bio, Inc.
Figures











References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

