Effects of Fructose and Stress on Rat Renal Copper Metabolism and Antioxidant Enzymes Function
- PMID: 36012287
- PMCID: PMC9409054
- DOI: 10.3390/ijms23169023
Effects of Fructose and Stress on Rat Renal Copper Metabolism and Antioxidant Enzymes Function
Abstract
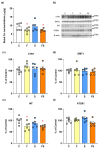
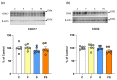
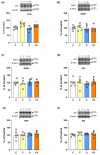
The effects of a fructose-rich diet and chronic stress on copper metabolism in the kidneys are still understudied. We investigated whether fructose and/or chronic unpredictable stress modulate copper metabolism in a way that affects redox homeostasis, thus contributing to progression of metabolic disturbances in the kidney. We determined protein level of copper transporters, chaperones, and cuproenzymes including cytochrome c oxidase, as well as antioxidant enzymes function in the kidneys of male Wistar rats subjected to 20% liquid fructose supplementation and/or chronic stress. Liquid fructose supplementation increased level of copper chaperone of superoxide dismutase and decreased metallothionein level, while rendering the level of copper importer and copper chaperones involved in copper delivery to mitochondria and trans Golgi network unaffected. Stress had no effect on renal copper metabolism. The activity and expression of renal antioxidant enzymes remained unaltered in all experimental groups. In conclusion, fructose, independently of stress, decreased renal copper level, and modulated renal copper metabolism as to preserve vital cellular function including mitochondrial energy production and antioxidative defense, at the expense of intracellular copper storage.
Keywords: copper chaperone of superoxide dismutase; copper transporter; fructose-fed rat; kidney; oxidative stress.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures
Similar articles
-
Diphenyl diselenide supplementation reduces biochemical alterations associated with oxidative stress in rats fed with fructose and hydrochlorothiazide.Chem Biol Interact. 2013 Aug 25;204(3):191-9. doi: 10.1016/j.cbi.2013.05.008. Epub 2013 May 22. Chem Biol Interact. 2013. PMID: 23707192
-
Chronic Stress Combined with a Fructose Diet Reduces Hypothalamic Insulin Signaling and Antioxidative Defense in Female Rats.Neuroendocrinology. 2019;108(4):278-290. doi: 10.1159/000496391. Epub 2018 Dec 20. Neuroendocrinology. 2019. PMID: 30572328
-
The expression and activity of antioxidant enzymes in the liver of rats exposed to high-fructose diet in the period from weaning to adulthood.J Sci Food Agric. 2015 Aug 30;95(11):2319-24. doi: 10.1002/jsfa.6953. Epub 2014 Nov 10. J Sci Food Agric. 2015. PMID: 25307280
-
Metal ion homeostasis with emphasis on zinc and copper: Potential crucial link to explain the non-classical antioxidative properties of vitamin D and melatonin.Life Sci. 2021 Sep 15;281:119770. doi: 10.1016/j.lfs.2021.119770. Epub 2021 Jun 28. Life Sci. 2021. PMID: 34197883 Review.
-
Cardiovascular Harmful Effects of Recommended Daily Doses (13 µg/kg/day), Tolerable Upper Intake Doses (0.14 mg/kg/day) and Twice the Tolerable Doses (0.28 mg/kg/day) of Copper.Cardiovasc Toxicol. 2023 Jun;23(5-6):218-229. doi: 10.1007/s12012-023-09797-3. Epub 2023 May 30. Cardiovasc Toxicol. 2023. PMID: 37254026 Review.
Cited by
-
Effect of Copper on the Function of Isolated Porcine Kidneys Stored Using Simple Hypothermia.Int J Mol Sci. 2022 Oct 27;23(21):13031. doi: 10.3390/ijms232113031. Int J Mol Sci. 2022. PMID: 36361819 Free PMC article.
-
Development of a novel copper metabolism-related risk model to predict prognosis and tumor microenvironment of patients with stomach adenocarcinoma.Front Pharmacol. 2023 May 22;14:1185418. doi: 10.3389/fphar.2023.1185418. eCollection 2023. Front Pharmacol. 2023. PMID: 37284310 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

