Curcumin-Based Nanoformulations: A Promising Adjuvant towards Cancer Treatment
- PMID: 36014474
- PMCID: PMC9414608
- DOI: 10.3390/molecules27165236
Curcumin-Based Nanoformulations: A Promising Adjuvant towards Cancer Treatment
Abstract
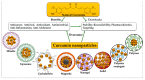
Throughout the United States, cancer remains the second leading cause of death. Traditional treatments induce significant medical toxic effects and unpleasant adverse reactions, making them inappropriate for long-term use. Consequently, anticancer-drug resistance and relapse are frequent in certain situations. Thus, there is an urgent necessity to find effective antitumor medications that are specific and have few adverse consequences. Curcumin is a polyphenol derivative found in the turmeric plant (Curcuma longa L.), and provides chemopreventive, antitumor, chemo-, and radio-sensitizing properties. In this paper, we summarize the new nano-based formulations of polyphenolic curcumin because of the growing interest in its application against cancers and tumors. According to recent studies, the use of nanoparticles can overcome the hydrophobic nature of curcumin, as well as improving its stability and cellular bioavailability in vitro and in vivo. Several strategies for nanocurcumin production have been developed, each with its own set of advantages and unique features. Because the majority of the curcumin-based nanoformulation evidence is still in the conceptual stage, there are still numerous issues impeding the provision of nanocurcumin as a possible therapeutic option. To support the science, further work is necessary to develop curcumin as a viable anti-cancer adjuvant. In this review, we cover the various curcumin nanoformulations and nanocurcumin implications for therapeutic uses for cancer, as well as the current state of clinical studies and patents. We further address the knowledge gaps and future research orientations required to develop curcumin as a feasible treatment candidate.
Keywords: cancer immunotherapy; cancer treatment; curcumin nanoformulations; drug delivery system; nanocarriers; nanocurcumin.
Conflict of interest statement
The authors declare no competing interest.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

