The role and mechanism of butyrate in the prevention and treatment of diabetic kidney disease
- PMID: 36016798
- PMCID: PMC9396028
- DOI: 10.3389/fmicb.2022.961536
The role and mechanism of butyrate in the prevention and treatment of diabetic kidney disease
Abstract
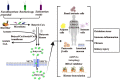
Diabetic kidney disease (DKD) remains the leading cause of the end-stage renal disease and is a major burden on the healthcare system. The current understanding of the mechanisms responsible for the progression of DKD recognizes the involvement of oxidative stress, low-grade inflammation, and fibrosis. Several circulating metabolites that are the end products of the fermentation process, released by the gut microbiota, are known to be associated with systemic immune-inflammatory responses and kidney injury. This phenomenon has been recognized as the "gut-kidney axis." Butyrate is produced predominantly by gut microbiota fermentation of dietary fiber and undigested carbohydrates. In addition to its important role as a fuel for colonic epithelial cells, butyrate has been demonstrated to ameliorate obesity, diabetes, and kidney diseases via G-protein coupled receptors (GPCRs). It also acts as an epigenetic regulator by inhibiting histone deacetylase (HDAC), up-regulation of miRNAs, or induction of the histone butyrylation and autophagy processes. This review aims to outline the existing literature on the treatment of DKD by butyrate in animal models and cell culture experiments, and to explore the protective effects of butyrate on DKD and the underlying molecular mechanism.
Keywords: butyrate; diabetic kidney disease; epigenetics; immune; inflammation.
Copyright © 2022 Cheng, Zhou, He, Xie, Xu and Huang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources

