Structural aspects of chemical modifications in the MHC-restricted immunopeptidome; Implications for immune recognition
- PMID: 36017166
- PMCID: PMC9395651
- DOI: 10.3389/fchem.2022.861609
Structural aspects of chemical modifications in the MHC-restricted immunopeptidome; Implications for immune recognition
Abstract
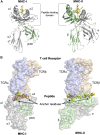
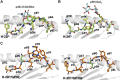
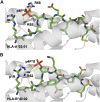
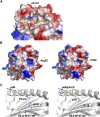
Significant advances in mass-spectroscopy (MS) have made it possible to investigate the cellular immunopeptidome, a large collection of MHC-associated epitopes presented on the surface of healthy, stressed and infected cells. These approaches have hitherto allowed the unambiguous identification of large cohorts of epitope sequences that are restricted to specific MHC class I and II molecules, enhancing our understanding of the quantities, qualities and origins of these peptide populations. Most importantly these analyses provide essential information about the immunopeptidome in responses to pathogens, autoimmunity and cancer, and will hopefully allow for future tailored individual therapies. Protein post-translational modifications (PTM) play a key role in cellular functions, and are essential for both maintaining cellular homeostasis and increasing the diversity of the proteome. A significant proportion of proteins is post-translationally modified, and thus a deeper understanding of the importance of PTM epitopes in immunopeptidomes is essential for a thorough and stringent understanding of these peptide populations. The aim of the present review is to provide a structural insight into the impact of PTM peptides on stability of MHC/peptide complexes, and how these may alter/modulate immune responses.
Keywords: T cell receptor (TCR); epitope; human leucocyte antigens (HLA); immune responses; immunopeptidome; major histocompatibility complex (MHC); post-translation modification.
Copyright © 2022 Sandalova, Sala and Achour.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Critical Review of Existing MHC I Immunopeptidome Isolation Methods.Molecules. 2020 Nov 19;25(22):5409. doi: 10.3390/molecules25225409. Molecules. 2020. PMID: 33228004 Free PMC article. Review.
-
Digging deeper into the immunopeptidome: characterization of post-translationally modified peptides presented by MHC I.J Proteins Proteom. 2021;12(3):151-160. doi: 10.1007/s42485-021-00066-x. Epub 2021 Jun 4. J Proteins Proteom. 2021. PMID: 36619276 Free PMC article.
-
Quantification of HLA-DM-Dependent Major Histocompatibility Complex of Class II Immunopeptidomes by the Peptide Landscape Antigenic Epitope Alignment Utility.Front Immunol. 2018 May 3;9:872. doi: 10.3389/fimmu.2018.00872. eCollection 2018. Front Immunol. 2018. PMID: 29774024 Free PMC article.
-
Contemplating immunopeptidomes to better predict them.Semin Immunol. 2023 Mar;66:101708. doi: 10.1016/j.smim.2022.101708. Epub 2023 Jan 6. Semin Immunol. 2023. PMID: 36621290 Review.
-
Post-translational modifications reshape the antigenic landscape of the MHC I immunopeptidome in tumors.Nat Biotechnol. 2023 Feb;41(2):239-251. doi: 10.1038/s41587-022-01464-2. Epub 2022 Oct 6. Nat Biotechnol. 2023. PMID: 36203013 Free PMC article.
Cited by
-
APE-Gen2.0: Expanding Rapid Class I Peptide-Major Histocompatibility Complex Modeling to Post-Translational Modifications and Noncanonical Peptide Geometries.J Chem Inf Model. 2024 Mar 11;64(5):1730-1750. doi: 10.1021/acs.jcim.3c01667. Epub 2024 Feb 28. J Chem Inf Model. 2024. PMID: 38415656 Free PMC article.
-
Engineered Proteins and Chemical Tools to Probe the Cell Surface Proteome.Chem Rev. 2025 Apr 23;125(8):4069-4110. doi: 10.1021/acs.chemrev.4c00554. Epub 2025 Apr 3. Chem Rev. 2025. PMID: 40178992 Free PMC article. Review.
-
Exploring the dynamic landscape of immunopeptidomics: Unravelling posttranslational modifications and navigating bioinformatics terrain.Mass Spectrom Rev. 2025 Jul-Aug;44(4):599-629. doi: 10.1002/mas.21905. Epub 2024 Aug 16. Mass Spectrom Rev. 2025. PMID: 39152539 Free PMC article. Review.
-
A Chemical Approach to Assess the Impact of Post-translational Modification on MHC Peptide Binding and Effector Cell Engagement.ACS Chem Biol. 2024 Sep 20;19(9):1991-2001. doi: 10.1021/acschembio.4c00312. Epub 2024 Aug 16. ACS Chem Biol. 2024. PMID: 39150956 Free PMC article.
-
Profound N-glycan remodelling accompanies MHC-II immunopeptide presentation.Front Immunol. 2023 Nov 8;14:1258518. doi: 10.3389/fimmu.2023.1258518. eCollection 2023. Front Immunol. 2023. PMID: 38022636 Free PMC article.
References
-
- Achour A., Michaëlsson J., Harris R. A., Odeberg J., Grufman P., Sandberg J. K., et al. (2002). A structural basis for LCMV immune evasion: Subversion of H-2D(b) and H-2K(b) presentation of gp33 revealed by comparative crystal structure.Analyses. Immunity 17, 757–768. 10.1016/s1074-7613(02)00478-8 - DOI - PubMed
-
- Andersen M. H., Bonfill J. E., Neisig A., Arsequell G., Sondergaard I., Valencia G., et al. (1999). Phosphorylated peptides can be transported by TAP molecules, presented by class I MHC molecules, and recognized by phosphopeptide-specific CTL. J. Immunol. 163, 3812–3818. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

