The impact of noradrenergic neurotoxin DSP-4 and noradrenaline transporter knockout (NET-KO) on the activity of liver cytochrome P450 3A (CYP3A) in male and female mice
- PMID: 36018449
- PMCID: PMC9584982
- DOI: 10.1007/s43440-022-00406-8
The impact of noradrenergic neurotoxin DSP-4 and noradrenaline transporter knockout (NET-KO) on the activity of liver cytochrome P450 3A (CYP3A) in male and female mice
Abstract
Background: Our earlier studies have shown that the brain noradrenergic system regulates cytochrome P450 (CYP) in rat liver via neuroendocrine mechanism. In the present work, a comparative study on the effect of intraperitoneal administration of the noradrenergic neurotoxin DSP-4 and the knockout of noradrenaline transporter (NET-KO) on the CYP3A in the liver of male and female mice was performed.
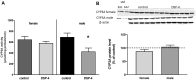
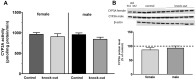
Methods: The experiments were conducted on C57BL/6J WT and NET-/- male/female mice. DSP-4 was injected intraperitoneally as a single dose (50 mg/kg ip.) to WT mice. The activity of CYP3A was measured as the rate of 6β-hydroxylation of testosterone in liver microsomes. The CYP3A protein level was estimated by Western blotting.
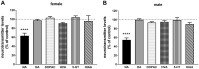
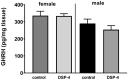
Results: DSP-4 evoked a selective decrease in the noradrenaline level in the brain of male and female mice. At the same time, DSP-4 reduced the CYP3A activity in males, but not in females. The level of CYP3A protein was not changed. The NET knockout did not affect the CYP3A activity/protein in both sexes.
Conclusions: The results with DSP-4 treated mice showed sex-dependent differences in the regulation of liver CYP3A by the brain noradrenergic system (with only males being responsive), and revealed that the NET knockout did not affect CYP3A in both sexes. Further studies into the hypothalamic-pituitary-gonadal hormones in DSP-4 treated mice may explain sex-specific differences in CYP3A regulation, whereas investigation of monoaminergic receptor sensitivity in the hypothalamic/pituitary areas of NET-/- mice will allow for understanding a lack of changes in the CYP3A activity in the NET-KO animals.
Keywords: Brain; Cytochrome P450; DSP-4 neurotoxin; Liver; NET knockout; Neuroendocrine regulation; Noradrenergic system.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures
Similar articles
-
The role of brain noradrenergic system in the regulation of liver cytochrome P450 expression.Biochem Pharmacol. 2013 Sep 15;86(6):800-7. doi: 10.1016/j.bcp.2013.07.017. Epub 2013 Aug 4. Biochem Pharmacol. 2013. PMID: 23924605
-
Involvement of the paraventricular (PVN) and arcuate (ARC) nuclei of the hypothalamus in the central noradrenergic regulation of liver cytochrome P450.Biochem Pharmacol. 2013 Dec 1;86(11):1614-20. doi: 10.1016/j.bcp.2013.09.006. Epub 2013 Sep 17. Biochem Pharmacol. 2013. PMID: 24051135
-
Cytochrome P450 is regulated by noradrenergic and serotonergic systems.Pharmacol Res. 2011 Oct;64(4):371-80. doi: 10.1016/j.phrs.2011.06.020. Epub 2011 Jun 30. Pharmacol Res. 2011. PMID: 21742037
-
Gender-dependent activity of CYP3A is indirectly modified by GR in the noradrenergic system.Pharmacol Rep. 2013;65(5):1431-4. doi: 10.1016/s1734-1140(13)71503-2. Pharmacol Rep. 2013. PMID: 24399741
-
The role of the nervous system in the regulation of liver cytochrome p450.Curr Drug Metab. 2011 Feb;12(2):124-38. doi: 10.2174/138920011795016908. Curr Drug Metab. 2011. PMID: 21401511 Review.
Cited by
-
Genetic loss of norepinephrine does not alter adult hippocampal neurogenesis in dopamine beta-hydroxylase deficient mice.IBRO Neurosci Rep. 2022 Oct 27;13:420-425. doi: 10.1016/j.ibneur.2022.10.010. eCollection 2022 Dec. IBRO Neurosci Rep. 2022. PMID: 36386600 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
