Attractor-like Dynamics in the Subicular Complex
- PMID: 36028315
- PMCID: PMC9546466
- DOI: 10.1523/JNEUROSCI.2048-20.2022
Attractor-like Dynamics in the Subicular Complex
Abstract
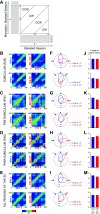
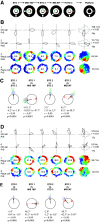
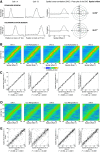
Distinct computations are performed at multiple brain regions during the encoding of spatial environments. Neural representations in the hippocampal, entorhinal, and head direction (HD) networks during spatial navigation have been clearly documented, while the representational properties of the subicular complex (SC) are relatively underexplored, although it has extensive anatomic connections with various brain regions involved in spatial information processing. We simultaneously recorded single units from different subregions of the SC in male rats while they ran clockwise on a centrally placed textured circular track (four different textures, each covering a quadrant), surrounded by six distal cues. The neural activity was monitored in standard sessions by maintaining the same configuration between the cues, while in cue manipulation sessions, the distal and local cues were either rotated in opposite directions to create a mismatch between them or the distal cues were removed. We report a highly coherent neural representation of the environment and a robust coupling between the HD cells and the spatial cells in the SC, strikingly different from previous reports of coupling between cells from co-recorded sites. Neural representations were (1) originally governed by the distal cues under local-distal cue-conflict conditions, (2) controlled by the local cues in the absence of distal cues, and (3) governed by the cues that are perceived to be stable. We propose that such attractor-like dynamics in the SC might play a critical role in the orientation of spatial representations, thus providing a "reference map" of the environment for further processing by other networks.SIGNIFICANCE STATEMENT The subicular complex (SC) receives major inputs from the entorhinal cortex and the hippocampus, and head direction (HD) information directly from the HD system. Using cue-conflict experiments, we studied the hierarchical representation of the local and distal cues in the SC to understand its role in the cognitive map, and report a highly coherent neural representation with robust coupling between the HD cells and the spatial cells in different subregions of the SC exhibiting attractor-like dynamics unaffected by the cue manipulations, strikingly different from previous reports of coupling between cells from co-recorded sites. This unique feature may allow the SC to function as a single computational unit during the representation of space, which may serve as a reference map of the environment.
Keywords: attractor dynamics; head direction cell; neural representation; place cell; spatial navigation; subicular complex.
Copyright © 2022 the authors.
Figures



Similar articles
-
Head direction cell representations maintain internal coherence during conflicting proximal and distal cue rotations: comparison with hippocampal place cells.J Neurosci. 2006 Jan 11;26(2):622-31. doi: 10.1523/JNEUROSCI.3885-05.2006. J Neurosci. 2006. PMID: 16407560 Free PMC article.
-
Distal CA1 Maintains a More Coherent Spatial Representation than Proximal CA1 When Local and Global Cues Conflict.J Neurosci. 2021 Nov 24;41(47):9767-9781. doi: 10.1523/JNEUROSCI.2938-20.2021. Epub 2021 Oct 20. J Neurosci. 2021. PMID: 34670850 Free PMC article.
-
Hippocampus Maintains a Coherent Map Under Reward Feature-Landmark Cue Conflict.Front Neural Circuits. 2022 Apr 26;16:878046. doi: 10.3389/fncir.2022.878046. eCollection 2022. Front Neural Circuits. 2022. PMID: 35558552 Free PMC article.
-
Tracking the flow of hippocampal computation: Pattern separation, pattern completion, and attractor dynamics.Neurobiol Learn Mem. 2016 Mar;129:38-49. doi: 10.1016/j.nlm.2015.10.008. Epub 2015 Oct 26. Neurobiol Learn Mem. 2016. PMID: 26514299 Free PMC article. Review.
-
Entorhinal-hippocampal interactions lead to globally coherent representations of space.Curr Res Neurobiol. 2022 Mar 21;3:100035. doi: 10.1016/j.crneur.2022.100035. eCollection 2022. Curr Res Neurobiol. 2022. PMID: 36685760 Free PMC article. Review.
Cited by
-
Historical loss weakens competitive behavior by remodeling ventral hippocampal dynamics.Cell Discov. 2025 Feb 25;11(1):16. doi: 10.1038/s41421-024-00751-3. Cell Discov. 2025. PMID: 39994206 Free PMC article.
-
Environment geometry alters subiculum boundary vector cell receptive fields in adulthood and early development.Nat Commun. 2024 Feb 1;15(1):982. doi: 10.1038/s41467-024-45098-1. Nat Commun. 2024. PMID: 38302455 Free PMC article.
References
-
- Berens P (2009) CircStat: a MATLAB Toolbox for circular statistics. J Stat Softw 31:1–21.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous
