mRNA vaccines in the prevention and treatment of diseases
- PMID: 36033422
- PMCID: PMC9409637
- DOI: 10.1002/mco2.167
mRNA vaccines in the prevention and treatment of diseases
Abstract
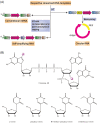
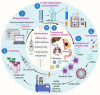
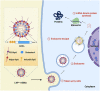
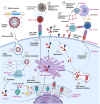
Messenger ribonucleic acid (mRNA) vaccines made their successful public debut in the effort against the COVID-19 outbreak starting in late 2019, although the history of mRNA vaccines can be traced back decades. This review provides an overview to discuss the historical course and present situation of mRNA vaccine development in addition to some basic concepts that underly mRNA vaccines. We discuss the general preparation and manufacturing of mRNA vaccines and also discuss the scientific advances in the in vivo delivery system and evaluate popular approaches (i.e., lipid nanoparticle and protamine) in detail. Next, we highlight the clinical value of mRNA vaccines as potent candidates for therapeutic treatment and discuss clinical progress in the treatment of cancer and coronavirus disease 2019. Data suggest that mRNA vaccines, with several prominent advantages, have achieved encouraging results and increasing attention due to tremendous potential in disease management. Finally, we suggest some potential directions worthy of further investigation and optimization. In addition to basic research, studies that help to facilitate storage and transportation will be indispensable for practical applications.
Keywords: cancer; infectious diseases; lipid nanoparticles; mRNA delivery; mRNA vaccine.
© 2022 The Authors. MedComm published by Sichuan International Medical Exchange & Promotion Association (SCIMEA) and John Wiley & Sons Australia, Ltd.
Conflict of interest statement
The authors declare there is no conflict of interest.
Figures

References
-
- Kardani K, Bolhassani AJC. Vaccine development against SARS‐CoV‐2: from virology to vaccine clinical trials. Coronaviruses. 2021;2(2):159‐171.
-
- Pardi N, Hogan MJ, Weissman D. Recent advances in mRNA vaccine technology. Curr Opin Immunol. 2020;65:14‐20. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
