Leishmania guyanensis suppressed inducible nitric oxide synthase provoked by its viral endosymbiont
- PMID: 36034693
- PMCID: PMC9416488
- DOI: 10.3389/fcimb.2022.944819
Leishmania guyanensis suppressed inducible nitric oxide synthase provoked by its viral endosymbiont
Abstract
Inducible nitric oxide synthase (iNOS) is essential to the production of nitric oxide (NO), an efficient effector molecule against intracellular human pathogens such as Leishmania protozoan parasites. Some strains of Leishmania are known to bear a viral endosymbiont termed Leishmania RNA virus 1 (LRV1). Recognition of LRV1 by the innate immune sensor Toll-like receptor-3 (TLR3) leads to conditions worsening the disease severity in mice. This process is governed by type I interferon (type I IFNs) arising downstream of TLR3 stimulation and favoring the formation of secondary metastatic lesions. The formation of these lesions is mediated by the inflammatory cytokine IL-17A and occurs in the absence, or low level of, protective cytokine IFN-γ. Here, we described that the presence of LRV1 led to the initial expression of iNOS and low production of NO that failed to control infection. We subsequently showed that LRV1-triggered type I IFN was essential but insufficient to induce robust iNOS induction, which requires strong activation of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB). Leishmania guyanensis carrying LRV1 (LgyLRV1+) parasites mitigated strong iNOS production by limiting NF-kB activation via the induction of tumor necrosis factor-alpha-induced protein 3 (TNFAIP3), also known as A20. Moreover, our data suggested that production of LRV1-induced iNOS could be correlated with parasite dissemination and metastasis via elevated secretion of IL-17A in the draining lymph nodes. Our findings support an additional strategy by which LRV1-bearing Leishmania guyanensis evaded killing by nitric oxide and suggest that low levels of LRV1-induced NO might contribute to parasite metastasis.
Keywords: IL-17A; Leishmania; Leishmania RNA virus 1 (LRV1); inducible nitric oxide synthase (iNOS); metastasis; nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kB); tumor necrosis factor-alpha-induced protein 3 (A20); type I Interferons.
Copyright © 2022 Kopelyanskiy, Desponds, Prevel, Rossi, Migliorini, Snäkä, Eren, Claudinot, Lye, Pasparakis, Beverley and Fasel.
Conflict of interest statement
The authors declare that the research was carried out in the absence of any commercial or financial relationships that could result in a potential conflict of interest.
Figures
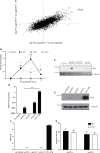


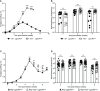


References
-
- Adaui V., Lye L.-F., Akopyants N. S., Zimic M., Llanos-Cuentas A., Garcia L., et al. . (2016). Association of the endobiont doubled-stranded RNA virus LRV1 with treatment failure of human leishmaniasis caused by leishmania braziliensis in Peru and Bolivia. J. Infect. Dis 213 (1), 112–21. doi: 10.1093/infdis/jiv354 - DOI - PMC - PubMed
-
- Aguirre-Garcia M. M., Escalona-Montaño A. R., Wilkins-Rodríguez A. A., Gutiérrez-Kobeh L. (2018). Immune evasion strategies. Leishmaniases Re-emerg. Dis. (Chapter 4: Innate Immune Evasion Strategies) doi: 10.5772/intechopen.75169 - DOI
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

