Microbial Aerosols Generated from Standard Microbiological Laboratory Procedures
- PMID: 36035500
- PMCID: PMC9150131
- DOI: 10.1089/apb.2021.0038
Microbial Aerosols Generated from Standard Microbiological Laboratory Procedures
Abstract
Background: Modern microbiology laboratories are designed to protect workers and the environment from microbial aerosols produced during microbiological procedures and accidents. However, there is only limited data available on the aerosols generated from common microbiology procedures.
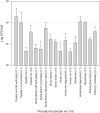
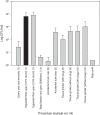
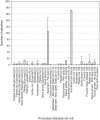
Methods: A series of common microbiological procedures were undertaken with high concentration spore suspensions while air samplers were operated to sample the aerosols generated. Surface contamination from droplets was visualized using sodium fluorescein within the suspension. A total of 36 procedures were studied using different sample volumes (0.1-10 mL) and two spore suspension titers (107 and 109 colony forming units [cfu]/mL).
Results: The aerosol concentrations generated varied from 0 to 13,000 cfu/m3. There was evidence to suggest that titer, volume, and poor use of equipment were significant factors in increased aerosol generation from some of the procedures. A risk assessment undertaken using the data showed that any aerosol generated from these processes would be contained within a correctly operating biological safety cabinet. Therefore, with these procedures, the operator and the environment would not require any additional protective measures such as respiratory protective equipment or a negative pressure laboratory to prevent aerosol exposure or release.
Conclusions: Aerosol generation from common laboratory processes can be minimized by reducing sample volumes and concentrations if possible. Training laboratory staff in good microbiological techniques would further mitigate aerosols generated from common laboratory processes.
Keywords: aerosol generation; bacterial spore; laboratory procedures; pipetting; spillage.
© Thomas Pottage et al. 2022; Published by Mary Ann Liebert, Inc.
Conflict of interest statement
No competing financial interests exist.
Figures

References
-
- World Health Organisation. Laboratory Biosafety Manual, 4th ed. Geneva: World Health Organization, 2020.
-
- US Department of Health and Human Services PHS, Centers for Disease Control and Prevention, National Institutes of Health. Biosafety in Microbiological and Biomedical Laboratories, 6th ed; 2020. USA: US Department of Health and Human Services.
-
- British Standard Institute. BS EN 12469:2000 Biotechnology. Performance Criteria for Microbiological Safety Cabinets. United Kingdom: British Standards Institute, 2000.
-
- Kojima K, Booth CM, Summermatter K, et al. Risk-based reboot for global lab biosafety. Science. 2018;360(6386):260–262. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
