Quality control mechanisms that protect nuclear envelope identity and function
- PMID: 36036741
- PMCID: PMC9442147
- DOI: 10.1083/jcb.202205123
Quality control mechanisms that protect nuclear envelope identity and function
Abstract
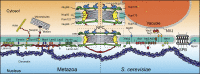
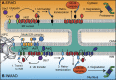
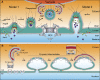
The nuclear envelope (NE) is a specialization of the endoplasmic reticulum with distinct biochemistry that defines inner and outer membranes connected at a pore membrane that houses nuclear pore complexes (NPCs). Quality control mechanisms that maintain the physical integrity and biochemical identity of these membranes are critical to ensure that the NE acts as a selective barrier that also contributes to genome stability and metabolism. As the proteome of the NE is highly integrated, it is challenging to turn over by conventional ubiquitin-proteasome and autophagy mechanisms. Further, removal of entire sections of the NE requires elaborate membrane remodeling that is poorly understood. Nonetheless, recent work has made inroads into discovering specializations of cellular degradative machineries tailored to meeting the unique challenges imposed by the NE. In addition, cells have evolved mechanisms to surveil and repair the NE barrier to protect against the deleterious effects of a breach in NE integrity, in the form of either a ruptured NE or a dysfunctional NPC. Here, we synthesize the most recent work exploring NE quality control mechanisms across eukaryotes.
© 2022 Mannino and Lusk.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

