Acid sphingomyelinase deactivation post-ischemia promotes brain angiogenesis and remodeling by small extracellular vesicles
- PMID: 36038749
- PMCID: PMC9424180
- DOI: 10.1007/s00395-022-00950-7
Acid sphingomyelinase deactivation post-ischemia promotes brain angiogenesis and remodeling by small extracellular vesicles
Abstract
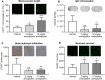
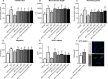
Antidepressants have been reported to enhance stroke recovery independent of the presence of depressive symptoms. They have recently been proposed to exert their mood-stabilizing actions by inhibition of acid sphingomyelinase (ASM), which catalyzes the hydrolysis of sphingomyelin to ceramide. Their restorative action post-ischemia/reperfusion (I/R) still had to be defined. Mice subjected to middle cerebral artery occlusion or cerebral microvascular endothelial cells exposed to oxygen-glucose deprivation were treated with vehicle or with the chemically and pharmacologically distinct antidepressants amitriptyline, fluoxetine or desipramine. Brain ASM activity significantly increased post-I/R, in line with elevated ceramide levels in microvessels. ASM inhibition by amitriptyline reduced ceramide levels, and increased microvascular length and branching point density in wildtype, but not sphingomyelinase phosphodiesterase-1 ([Smpd1]-/-) (i.e., ASM-deficient) mice, as assessed by 3D light sheet microscopy. In cell culture, amitriptyline, fluoxetine, and desipramine increased endothelial tube formation, migration, VEGFR2 abundance and VEGF release. This effect was abolished by Smpd1 knockdown. Mechanistically, the promotion of angiogenesis by ASM inhibitors was mediated by small extracellular vesicles (sEVs) released from endothelial cells, which exhibited enhanced uptake in target cells. Proteomic analysis of sEVs revealed that ASM deactivation differentially regulated proteins implicated in protein export, focal adhesion, and extracellular matrix interaction. In vivo, the increased angiogenesis was accompanied by a profound brain remodeling response with increased blood-brain barrier integrity, reduced leukocyte infiltrates and increased neuronal survival. Antidepressive drugs potently boost angiogenesis in an ASM-dependent way. The release of sEVs by ASM inhibitors disclosed an elegant target, via which brain remodeling post-I/R can be amplified.
Keywords: Antidepressants; Ceramide; Exosome; Focal cerebral ischemia; Sphingomyelin; Stroke recovery.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures






References
-
- Abe T, Niizuma K, Kanoke A, Saigusa D, Saito R, Uruno A, Fujimura M, Yamamoto M, Tominaga T. Metabolomic Analysis of Mouse Brain after a Transient Middle Cerebral Artery Occlusion by Mass Spectrometry Imaging. Neurol Med Chir (Tokyo) 2018;58:384–392. doi: 10.2176/nmc.oa.2018-0054. - DOI - PMC - PubMed
-
- Benjamin EJ, Muntner P, Alonso A, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Chang AR, Cheng S, Das SR, Delling FN, Djousse L, Elkind MSV, Ferguson JF, Fornage M, Jordan LC, Khan SS, Kissela BM, Knutson KL, Kwan TW, Lackland DT, Lewis TT, Lichtman JH, Longenecker CT, Loop MS, Lutsey PL, Martin SS, Matsushita K, Moran AE, Mussolino ME, O'Flaherty M, Pandey A, Perak AM, Rosamond WD, Roth GA, Sampson UKA, Satou GM, Schroeder EB, Shah SH, Spartano NL, Stokes A, Tirschwell DL, Tsao CW, Turakhia MP, VanWagner LB, Wilkins JT, Wong SS, Virani SS, American Heart Association Council on E, Prevention Statistics C, Stroke Statistics S Heart Disease and stroke statistics-2019 update: a report from the American Heart Association. Circulation. 2019;139:e56–e528. doi: 10.1161/CIR.0000000000000659. - DOI - PubMed
-
- Brunkhorst R, Friedlaender F, Ferreiros N, Schwalm S, Koch A, Grammatikos G, Toennes S, Foerch C, Pfeilschifter J, Pfeilschifter W. Alterations of the ceramide metabolism in the peri-infarct cortex are independent of the sphingomyelinase pathway and not influenced by the acid sphingomyelinase inhibitor fluoxetine. Neural Plast. 2015 doi: 10.1155/2015/503079. - DOI - PMC - PubMed
-
- Chabriat H, Bassetti CL, Marx U, Audoli-Inthavong ML, Sors A, Lambert E, Wattez M, Hermann DM, investigators RBs Safety and efficacy of GABAA alpha5 antagonist S44819 in patients with ischaemic stroke: a multicentre, double-blind, randomised, placebo-controlled trial. Lancet Neurol. 2020;19:226–233. doi: 10.1016/S1474-4422(20)30004-1. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

