Tracking of Stem Cells from Human Exfoliated Deciduous Teeth Labeled with Molday ION Rhodamine-B during Periodontal Bone Regeneration in Rats
- PMID: 36042010
- PMCID: PMC9978830
- DOI: 10.15283/ijsc21204
Tracking of Stem Cells from Human Exfoliated Deciduous Teeth Labeled with Molday ION Rhodamine-B during Periodontal Bone Regeneration in Rats
Abstract
Background and objectives: Chronic periodontitis can lead to alveolar bone resorption and eventually tooth loss. Stem cells from exfoliated deciduous teeth (SHED) are appropriate bone regeneration seed cells. To track the survival, migration, and differentiation of the transplanted SHED, we used super paramagnetic iron oxide particles (SPIO) Molday ION Rhodamine-B (MIRB) to label and monitor the transplanted cells while repairing periodontal bone defects.
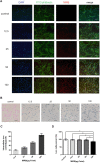
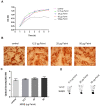
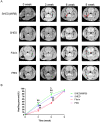
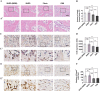
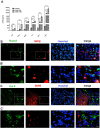
Methods and results: We determined an appropriate dose of MIRB for labeling SHED by examining the growth and osteogenic differentiation of labeled SHED. Finally, SHED was labeled with 25 μg Fe/ml MIRB before being transplanted into rats. Magnetic resonance imaging was used to track SHED survival and migration in vivo due to a low-intensity signal artifact caused by MIRB. HE and immunohistochemical analyses revealed that both MIRB-labeled and unlabeled SHED could promote periodontal bone regeneration. The colocalization of hNUC and MIRB demonstrated that SHED transplanted into rats could survive in vivo. Furthermore, some MIRB-positive cells expressed the osteoblast and osteocyte markers OCN and DMP1, respectively. Enzyme-linked immunosorbent assay revealed that SHED could secrete protein factors, such as IGF-1, OCN, ALP, IL-4, VEGF, and bFGF, which promote bone regeneration. Immunofluorescence staining revealed that the transplanted SHED was surrounded by a large number of host-derived Runx2- and Col II-positive cells that played important roles in the bone healing process.
Conclusions: SHED could promote periodontal bone regeneration in rats, and the survival of SHED could be tracked in vivo by labeling them with MIRB. SHED are likely to promote bone healing through both direct differentiation and paracrine mechanisms.
Keywords: Magnetic resonance imaging; Periodontal bone defect; Stem cells from human exfoliated deciduous teeth (SHED); Transplanted cells tracking.
Conflict of interest statement
The authors have no conflicting financial interest.
Figures
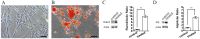


Similar articles
-
[Experimental study on transplantation of MIRB labeled human deciduous dental pulp stem cells to repair periodontal bone defects in rats].Shanghai Kou Qiang Yi Xue. 2022 Dec;31(6):581-587. Shanghai Kou Qiang Yi Xue. 2022. PMID: 36970792 Chinese.
-
Effects of the iron oxide nanoparticle Molday ION Rhodamine B on the viability and regenerative function of neural stem cells: relevance to clinical translation.Int J Nanomedicine. 2016 Apr 27;11:1731-48. doi: 10.2147/IJN.S102006. eCollection 2016. Int J Nanomedicine. 2016. PMID: 27175074 Free PMC article.
-
Biological Characteristics of Fluorescent Superparamagnetic Iron Oxide Labeled Human Dental Pulp Stem Cells.Stem Cells Int. 2017;2017:4837503. doi: 10.1155/2017/4837503. Epub 2017 Feb 16. Stem Cells Int. 2017. PMID: 28298928 Free PMC article.
-
Efficacy of Mesenchymal Stem Cells from Human Exfoliated Deciduous Teeth and their Derivatives in Inflammatory Diseases Therapy.Curr Stem Cell Res Ther. 2022;17(4):302-316. doi: 10.2174/1574888X17666220417153309. Curr Stem Cell Res Ther. 2022. PMID: 35440314 Review.
-
Expression of Odontogenic and Osteogenic Markers in DPSCs and SHED: A Review.Curr Stem Cell Res Ther. 2017;12(1):71-79. doi: 10.2174/1574888x11666160815095733. Curr Stem Cell Res Ther. 2017. PMID: 27527527 Review.
Cited by
-
The paradigm shifts of periodontal regeneration strategy: From reparative manipulation to developmental engineering.Bioact Mater. 2025 Mar 18;49:418-436. doi: 10.1016/j.bioactmat.2025.03.009. eCollection 2025 Jul. Bioact Mater. 2025. PMID: 40165829 Free PMC article. Review.
References
-
- Qiu J, Wang X, Zhou H, Zhang C, Wang Y, Huang J, Liu M, Yang P, Song A. Enhancement of periodontal tissue regeneration by conditioned media from gingiva-derived or periodontal ligament-derived mesenchymal stem cells: a comparative study in rats. Stem Cell Res Ther. 2020;11:42. doi: 10.1186/s13287-019-1546-9.07974615cc674308b98377718edb3909 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

