MicroRNA-375 is a therapeutic target for castration-resistant prostate cancer through the PTPN4/STAT3 axis
- PMID: 36042375
- PMCID: PMC9440249
- DOI: 10.1038/s12276-022-00837-6
MicroRNA-375 is a therapeutic target for castration-resistant prostate cancer through the PTPN4/STAT3 axis
Abstract
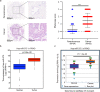
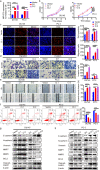
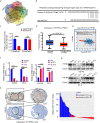
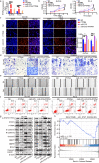
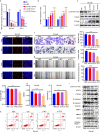
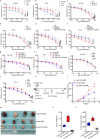
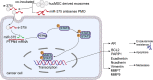
The functional role of microRNA-375 (miR-375) in the development of prostate cancer (PCa) remains controversial. Previously, we found that plasma exosomal miR-375 is significantly elevated in castration-resistant PCa (CRPC) patients compared with castration-sensitive PCa patients. Here, we aimed to determine how miR-375 modulates CRPC progression and thereafter to evaluate the therapeutic potential of human umbilical cord mesenchymal stem cell (hucMSC)-derived exosomes loaded with miR-375 antisense oligonucleotides (e-375i). We used miRNA in situ hybridization technique to evaluate miR-375 expression in PCa tissues, gain- and loss-of-function experiments to determine miR-375 function, and bioinformatic methods, dual-luciferase reporter assay, qPCR, IHC and western blotting to determine and validate the target as well as the effects of miR-375 at the molecular level. Then, e-375i complexes were assessed for their antagonizing effects against miR-375. We found that the expression of miR-375 was elevated in PCa tissues and cancer exosomes, correlating with the Gleason score. Forced expression of miR-375 enhanced the expression of EMT markers and AR but suppressed apoptosis markers, leading to enhanced proliferation, migration, invasion, and enzalutamide resistance and decreased apoptosis of PCa cells. These effects could be reversed by miR-375 silencing. Mechanistically, miR-375 directly interfered with the expression of phosphatase nonreceptor type 4 (PTPN4), which in turn stabilized phosphorylated STAT3. Application of e-375i could inhibit miR-375, upregulate PTPN4 and downregulate p-STAT3, eventually repressing the growth of PCa. Collectively, we identified a novel miR-375 target, PTPN4, that functions upstream of STAT3, and targeting miR-375 may be an alternative therapeutic for PCa, especially for CRPC with high AR levels.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Targeting Castration-Resistant Prostate Cancer Using Mesenchymal Stem Cell Exosomes for Therapeutic MicroRNA-let-7c Delivery.Front Biosci (Landmark Ed). 2022 Sep 6;27(9):256. doi: 10.31083/j.fbl2709256. Front Biosci (Landmark Ed). 2022. PMID: 36224011
-
GR silencing impedes the progression of castration-resistant prostate cancer through the JAG1/NOTCH2 pathway via up-regulation of microRNA-143-3p.Cancer Biomark. 2020;28(4):483-497. doi: 10.3233/CBM-191271. Cancer Biomark. 2020. PMID: 32568179 Free PMC article.
-
MicroRNA-144-3p inhibits cell proliferation and promotes apoptosis in castration-resistant prostate cancer by targeting CEP55.Eur Rev Med Pharmacol Sci. 2018 Nov;22(22):7660-7670. doi: 10.26355/eurrev_201811_16383. Eur Rev Med Pharmacol Sci. 2018. PMID: 30536308
-
Role of MicroRNA-21 in Prostate Cancer Progression and Metastasis: Molecular Mechanisms to Therapeutic Targets.Ann Surg Oncol. 2024 Jul;31(7):4795-4808. doi: 10.1245/s10434-024-15453-z. Epub 2024 May 17. Ann Surg Oncol. 2024. PMID: 38758485 Review.
-
[Progress in the studies of circular RNA in prostate cancer].Zhonghua Nan Ke Xue. 2022 Jul;28(7):649-655. Zhonghua Nan Ke Xue. 2022. PMID: 37556225 Review. Chinese.
Cited by
-
Nanoplatelets modified with RVG for targeted delivery of miR-375 and temozolomide to enhance gliomas therapy.J Nanobiotechnology. 2024 Oct 15;22(1):623. doi: 10.1186/s12951-024-02895-6. J Nanobiotechnology. 2024. PMID: 39402578 Free PMC article.
-
Mesenchymal Stem Cell-Derived Extracellular Vesicles in Cancer Therapy Resistance: from Biology to Clinical Opportunity.Int J Biol Sci. 2024 Jan 1;20(1):347-366. doi: 10.7150/ijbs.88500. eCollection 2024. Int J Biol Sci. 2024. PMID: 38164177 Free PMC article. Review.
-
MicroRNA-375 restrains the progression of lung squamous cell carcinoma by modulating the ERK pathway via UBE3A-mediated DUSP1 degradation.Cell Death Discov. 2023 Jun 29;9(1):199. doi: 10.1038/s41420-023-01499-7. Cell Death Discov. 2023. PMID: 37385985 Free PMC article.
-
State-of-the-art therapeutic strategies for targeting cancer stem cells in prostate cancer.Front Oncol. 2023 Mar 9;13:1059441. doi: 10.3389/fonc.2023.1059441. eCollection 2023. Front Oncol. 2023. PMID: 36969009 Free PMC article. Review.
-
KPNA5 Suppresses Malignant Progression of Ovarian Cancer Through Importing the PTPN4 Into the Nucleus.Cancer Med. 2025 Apr;14(7):e70731. doi: 10.1002/cam4.70731. Cancer Med. 2025. PMID: 40145330 Free PMC article.
References
-
- Sung H, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021;71:209–249. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

