Orthogonal Covalent Entrapment of Cargo into Biodegradable Polymeric Micelles via Native Chemical Ligation
- PMID: 36044412
- PMCID: PMC10565831
- DOI: 10.1021/acs.biomac.2c00865
Orthogonal Covalent Entrapment of Cargo into Biodegradable Polymeric Micelles via Native Chemical Ligation
Abstract
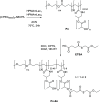
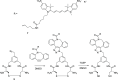
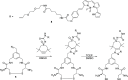
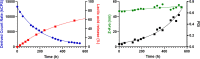
Polymeric micelles (PMs) are promising platforms for enhanced tissue targeting of entrapped therapeutic agents. Strategies to circumvent premature release of entrapped drugs include cross-linking of the micellar core as well as covalent attachment of the drug cargo. The chemistry employed to obtain cross-linked micelles needs to be mild to also allow entrapment of fragile molecules, such as certain peptides, proteins, oligonucleotides, and fluorescent dyes. Native chemical ligation (NCL) is a mild bio-orthogonal reaction between a N-terminal cysteine residue and a thioester that proceeds under physiological conditions. Here, we designed a trifunctional cross-linker containing two cysteine residues for the micelle core-cross-linking reaction and an azide residue for ring-strained alkyne conjugation of fluorescent dyes. We applied this approach to thermosensitive methoxypolyethylene glycol-b-N-(2-hydroxypropyl)methacrylamide-lactate (mPEG-b-HPMAmLacn) based block copolymers of a core-cross-linked polymeric micelle (CCPM) system by attaching thioester residues (using ethyl thioglycolate-succinic anhydride, ETSA) for NCL cross-linking with the trifunctional cross-linker under physiological conditions. By use of mild copper-free click chemistry, we coupled fluorescent dyes, Sulfo.Cy5 and BODIPY, to the core via the azide residue present on the cross-linker by triazole ring formation. In addition, we employed a recently developed cycloheptyne strain promoted click reagent (TMTHSI, CliCr) in comparison to the frequently employed cyclooctyne derivative (DBCO), both achieving successful dye entrapment. The size of the resulting CCPMs could be tuned between 50 and 100 nm by varying the molecular weight of the thermosensitive block and ETSA content. In vitro cell experiments showed successful internalization of the dye entrapped CCPMs, which did not affect cell viability up to a polymer concentration of 2 mg/mL in PC3 cells. These fluorescent dye entrapped CCPMs can be applied in diagnostic imaging and the chemistry developed in this study serves as a steppingstone toward covalently entrapped fragile drug compounds with tunable release in CCPMs.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




Similar articles
-
Mechanistic Study on the Degradation of Hydrolysable Core-Crosslinked Polymeric Micelles.Langmuir. 2023 Aug 29;39(34):12132-12143. doi: 10.1021/acs.langmuir.3c01399. Epub 2023 Aug 15. Langmuir. 2023. PMID: 37581242 Free PMC article.
-
Native Chemical Ligation for Cross-Linking of Flower-Like Micelles.Biomacromolecules. 2018 Sep 10;19(9):3766-3775. doi: 10.1021/acs.biomac.8b00908. Epub 2018 Aug 24. Biomacromolecules. 2018. PMID: 30102855 Free PMC article.
-
Versatile Click Linker Enabling Native Peptide Release from Nanocarriers upon Redox Trigger.Bioconjug Chem. 2023 Dec 20;34(12):2375-2386. doi: 10.1021/acs.bioconjchem.3c00484. Epub 2023 Dec 11. Bioconjug Chem. 2023. PMID: 38079189 Free PMC article.
-
Design, development and clinical translation of CriPec®-based core-crosslinked polymeric micelles.Adv Drug Deliv Rev. 2022 Dec;191:114613. doi: 10.1016/j.addr.2022.114613. Epub 2022 Nov 4. Adv Drug Deliv Rev. 2022. PMID: 36343757 Review.
-
Role of non-covalent and covalent interactions in cargo loading capacity and stability of polymeric micelles.J Control Release. 2014 Nov 10;193:9-26. doi: 10.1016/j.jconrel.2014.06.061. Epub 2014 Jul 15. J Control Release. 2014. PMID: 25037018 Review.
Cited by
-
Click Chemistry as an Efficient Toolbox for Coupling Sterically Hindered Molecular Systems to Obtain Advanced Materials for Nanomedicine.Int J Mol Sci. 2024 Dec 24;26(1):36. doi: 10.3390/ijms26010036. Int J Mol Sci. 2024. PMID: 39795895 Free PMC article. Review.
-
Mechanistic Study on the Degradation of Hydrolysable Core-Crosslinked Polymeric Micelles.Langmuir. 2023 Aug 29;39(34):12132-12143. doi: 10.1021/acs.langmuir.3c01399. Epub 2023 Aug 15. Langmuir. 2023. PMID: 37581242 Free PMC article.
References
-
- Varela-Moreira A.; Shi Y.; Fens M. H. A. M.; Lammers T.; Hennink W. E.; Schiffelers R. M. Clinical application of polymeric micelles for the treatment of cancer. Mater. Chem. Front. 2017, 1 (8), 1485–1501. 10.1039/C6QM00289G. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

