Phase Ia/b, Open-Label, Multicenter Study of AZD4635 (an Adenosine A2A Receptor Antagonist) as Monotherapy or Combined with Durvalumab, in Patients with Solid Tumors
- PMID: 36044531
- PMCID: PMC9660540
- DOI: 10.1158/1078-0432.CCR-22-0612
Phase Ia/b, Open-Label, Multicenter Study of AZD4635 (an Adenosine A2A Receptor Antagonist) as Monotherapy or Combined with Durvalumab, in Patients with Solid Tumors
Abstract
Purpose: To evaluate AZD4635, an adenosine A2A receptor antagonist, as monotherapy or in combination with durvalumab in patients with advanced solid tumors.
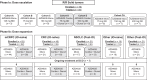
Patients and methods: In phase Ia (dose escalation), patients had relapsed/refractory solid tumors; in phase Ib (dose expansion), patients had checkpoint inhibitor-naïve metastatic castration-resistant prostate cancer (mCRPC) or colorectal carcinoma, non-small cell lung cancer with prior anti-PD-1/PD-L1 exposure, or other solid tumors (checkpoint-naïve or prior anti-PD-1/PD-L1 exposure). Patients received AZD4635 monotherapy (75-200 mg once daily or 125 mg twice daily) or in combination with durvalumab (AZD4635 75 or 100 mg once daily). The primary objective was safety; secondary objectives included antitumor activity and pharmacokinetics; exploratory objectives included evaluation of an adenosine gene signature in patients with mCRPC.
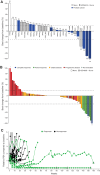
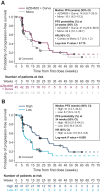
Results: As of September 8, 2020, 250 patients were treated (AZD4635, n = 161; AZD4635+durvalumab, n = 89). In phase Ia, DLTs were observed with monotherapy (125 mg twice daily; n = 2) and with combination treatment (75 mg; n = 1) in patients receiving nanosuspension. The most common treatment-related adverse events included nausea, fatigue, vomiting, decreased appetite, dizziness, and diarrhea. The RP2D of the AZD4635 capsule formulation was 75 mg once daily, as monotherapy or in combination with durvalumab. The pharmacokinetic profile was dose-proportional, and exposure was adequate to cover target with 100 mg nanosuspension or 75 mg capsule once daily. In patients with mCRPC receiving monotherapy or combination treatment, tumor responses (2/39 and 6/37, respectively) and prostate-specific antigen responses (3/60 and 10/45, respectively) were observed. High versus low blood-based adenosine signature was associated with median progression-free survival of 21 weeks versus 8.7 weeks.
Conclusions: AZD4635 monotherapy or combination therapy was well tolerated. Objective responses support additional phase II combination studies in patients with mCRPC.
©2022 The Authors; Published by the American Association for Cancer Research.
Figures

References
-
- Blay J, White TD, Hoskin DW. The extracellular fluid of solid carcinomas contains immunosuppressive concentrations of adenosine. Cancer Res 1997;57:2602–5. - PubMed
-
- Bendell J, Bauer T, Patel M, Falchook G, Karlix JL, Lim E, et al. Evidence of immune activation on the first-in-human phase 1a dose escalation study of the adenosine 2a receptor antagonist AZD4635, in patients with advanced solid tumors. Cancer Res 2019;79: 13s (suppl. abstr.CT026).
-
- Allard B, Allard D, Buisseret L, Stagg J. The adenosine pathway in immuno-oncology. Nat Rev Clin Oncol 2020;17:611–29. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

