Divergent clinical outcomes in a phase 2B trial of the TLPLDC vaccine in preventing melanoma recurrence and the impact of dendritic cell collection methodology: a randomized clinical trial
- PMID: 36045304
- PMCID: PMC9433518
- DOI: 10.1007/s00262-022-03272-8
Divergent clinical outcomes in a phase 2B trial of the TLPLDC vaccine in preventing melanoma recurrence and the impact of dendritic cell collection methodology: a randomized clinical trial
Abstract
Background: A randomized, double-blind, placebo-controlled phase 2b trial of the tumor lysate, particle-loaded, dendritic cell (TLPLDC) vaccine was conducted in patients with resected stage III/IV melanoma. Dendritic cells (DCs) were harvested with and without granulocyte-colony stimulating factor (G-CSF). This analysis investigates differences in clinical outcomes and RNA gene expression between DC harvest methods.
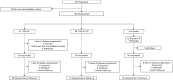
Methods: The TLPLDC vaccine is created by loading autologous tumor lysate into yeast cell wall particles (YCWPs) and exposing them to phagocytosis by DCs. For DC harvest, patients had a direct blood draw or were pretreated with G-CSF before blood draw. Patients were randomized 2:1 to receive TLPLDC or placebo. Differences in disease-free survival (DFS) and overall survival (OS) were evaluated. RNA-seq analysis was performed on the total RNA of TLPLDC + G and TLPLDC vaccines to compare gene expression between groups.
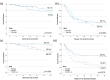
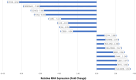
Results: 144 patients were randomized: 103 TLPLDC (47 TLPLDC/56 TLPLDC + G) and 41 placebo (19 placebo/22 placebo + G). Median follow-up was 27.0 months. Both 36-month DFS (55.8% vs. 24.4% vs. 30.0%, p = 0.010) and OS (94.2% vs. 69.8% vs. 70.9%, p = 0.024) were improved in TLPLDC compared to TLPLDC + G or placebo, respectively. When compared to TLPLDC + G vaccine, RNA-seq from TLPLDC vaccine showed upregulation of genes associated with DC maturation and downregulation of genes associated with DC suppression or immaturity.
Conclusions: Patients receiving TLPLDC vaccine without G-CSF had improved OS and DFS. Outcomes remained similar between patients receiving TLPLDC + G and placebo. Direct DC harvest without G-CSF had higher expression of genes linked to DC maturation, likely improving clinical efficacy.
Keywords: Cancer vaccine; Dendritic cell; Immunotherapy; Melanoma; Personalized medicine.
© 2022. The Author(s), under exclusive licence to Springer-Verlag GmbH Germany, part of Springer Nature.
Conflict of interest statement
The protocol was approved by the Western Institutional Review Board and all patients provided informed consent for participation. Dr. Faries is an advisor for Bristol-Myers Squibb, Sanofi, Array Bioscience and Pulse Bioscience. Dr. Wagner is an employee of Orbis Health Solutions. Dr. Peoples is employed by Orbis Health Solutions and Cancer Insight; is a consultant for Rapamycin Holdings, Heat Biologics, Abexxa Biologics, and Pelican Therapeutics; and has received funding from the above as well as Sellas Life Sciences and Genentech. Dr Jakub served on a Novartis Melanoma Surgical Oncology Advisory Board. Dr. Clifton is employed by Parthenon Therapeutics. The view(s) expressed herein are those of the author(s) and do not reflect the official policy or position of Brooke Army Medical Center, the US Army Medical Department, the Department of the Army, Department of the Air Force, Department of Defense, or the US Government. The voluntary, fully informed consent of the subjects used in this research was obtained as required by 32 CFR 219 and DODI 3216.02_AFI40-402.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

