Tumor Microenvironment and Immune Escape in the Time Course of Glioblastoma
- PMID: 36048342
- PMCID: PMC9525332
- DOI: 10.1007/s12035-022-02996-z
Tumor Microenvironment and Immune Escape in the Time Course of Glioblastoma
Abstract
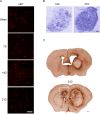
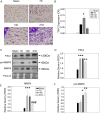
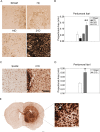
Glioblastoma multiforme (GBM) is the most aggressive primary brain tumor with a malignant prognosis. GBM is characterized by high cellular heterogeneity and its progression relies on the interaction with the central nervous system, which ensures the immune-escape and tumor promotion. This interplay induces metabolic, (epi)-genetic and molecular rewiring in both domains. In the present study, we aim to characterize the time-related changes in the GBM landscape, using a syngeneic mouse model of primary GBM. GL261 glioma cells were injected in the right striatum of immuno-competent C57Bl/6 mice and animals were sacrificed after 7, 14, and 21 days (7D, 14D, 21D). The tumor development was assessed through 3D tomographic imaging and brains were processed for immunohistochemistry, immunofluorescence, and western blotting. A human transcriptomic database was inquired to support the translational value of the experimental data. Our results showed the dynamic of the tumor progression, being established as a bulk at 14D and surrounded by a dense scar of reactive astrocytes. The GBM growth was paralleled by the impairment in the microglial/macrophagic recruitment and antigen-presenting functions, while the invasive phase was characterized by changes in the extracellular matrix, as shown by the analysis of tenascin C and metalloproteinase-9. The present study emphasizes the role of the molecular changes in the microenvironment during the GBM progression, fostering the development of novel multi-targeted, time-dependent therapies in an experimental model similar to the human disease.
Keywords: Astrocytes; FIB-2; Glioma; MHCII; MMP-9; Macrophages; Microglia; Neuroinflammation; Spatio-temporal heterogeneity.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

