Acoustic Force Elastography Microscopy
- PMID: 36049008
- PMCID: PMC9975118
- DOI: 10.1109/TBME.2022.3203435
Acoustic Force Elastography Microscopy
Abstract
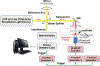
Objective: Hydrogel scaffolds have attracted attention to develop cellular therapy and tissue engineering platforms for regenerative medicine applications. Among factors, local mechanical properties of scaffolds drive the functionalities of cell niche. Dynamic mechanical analysis (DMA), the standard method to characterize mechanical properties of hydrogels, restricts development in tissue engineering because the measurement provides a single elasticity value for the sample, requires direct contact, and represents a destructive evaluation preventing longitudinal studies on the same sample. We propose a novel technique, acoustic force elastography microscopy (AFEM), to evaluate elastic properties of tissue engineering scaffolds.
Results: AFEM can resolve localized and two-dimensional (2D) elastic properties of both transparent and opaque materials with advantages of being non-contact and non-destructive. Gelatin hydrogels, neat synthetic oligo[poly(ethylene glycol)fumarate] (OPF) scaffolds, OPF hydroxyapatite nanocomposite scaffolds and ex vivo biological tissue were examined with AFEM to evaluate the elastic modulus. These measurements of Young's modulus range from approximately 2 kPa to over 100 kPa were evaluated and are in good agreement with finite element simulations, surface wave measurements, and DMA tests.
Conclusion: The AFEM can resolve localized and 2D elastic properties of hydrogels, scaffolds and thin biological tissues. These materials can either be transparent or non-transparent and their evaluation can be done in a non-contact and non-destructive manner, thereby facilitating longitudinal evaluation.
Significance: AFEM is a promising technique to quantify elastic properties of scaffolds for tissue engineering and will be applied to provide new insights for exploring elastic changes of cell-laden scaffolds for tissue engineering and material science.
Figures


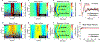






References
-
- Zhu Y et al., “Determination of mechanical properties of soft tissue scaffolds by atomic force microscopy nanoindentation,” J Biomech, vol. 44, no. 13, pp. 2356–61, 2011. - PubMed
-
- Fuentes E et al., “Structural characterization and mechanical performance of calcium phosphate scaffolds and natural bones: a comparative study,” J Appl Biomater Biomech, vol. 8, no. 3, pp. 159–65, 2010. - PubMed
-
- Rho JY et al., “Young’s modulus of trabecular and cortical bone material: ultrasonic and microtensile measurements,” J Biomech, vol. 26, no. 2, pp. 111–9, 1993. - PubMed

