Pulse oximetry for the diagnosis and management of acute respiratory distress syndrome
- PMID: 36049490
- PMCID: PMC9423770
- DOI: 10.1016/S2213-2600(22)00058-3
Pulse oximetry for the diagnosis and management of acute respiratory distress syndrome
Abstract
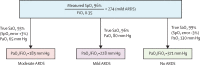
The diagnosis of acute respiratory distress syndrome (ARDS) traditionally requires calculation of the ratio of partial pressure of arterial oxygen to fraction of inspired oxygen (PaO2/FiO2) using arterial blood, which can be costly and is not possible in many resource-limited settings. By contrast, pulse oximetry is continuously available, accurate, inexpensive, and non-invasive. Pulse oximetry-based indices, such as the ratio of pulse-oximetric oxygen saturation to FiO2 (SpO2/FiO2), have been validated in clinical studies for the diagnosis and risk stratification of patients with ARDS. Limitations of the SpO2/FiO2 ratio include reduced accuracy in poor perfusion states or above oxygen saturations of 97%, and the potential for reduced accuracy in patients with darker skin pigmentation. Application of pulse oximetry to the diagnosis and management of ARDS, including formal adoption of the SpO2/FiO2 ratio as an alternative to PaO2/FiO2 to meet the diagnostic criterion for hypoxaemia in ARDS, could facilitate increased and earlier recognition of ARDS worldwide to advance both clinical practice and research.
Copyright © 2022 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of interests KDW has received research grant support from the US National Institutes of Health (NIH; 5T32GM008440-24). MAM has received research grant support from the NIH (HL134828, HL126456, HL140026, and 143896), the US Department of Defense, and Roche-Genentech for observational studies on acute respiratory distress syndrome; he reports consultancy fees from Citius Pharmaceuticals, Johnson & Johnson, Gilead Pharmaceuticals, Plant Therapeutics, and Novartis pharmaceuticals, outside of the submitted work. LBW has received research grant support from the NIH (HL103836, HL126176, HL158906) and the US Department of Defense, and grants or contracts from Genentech, Boehringer Ingelheim, and CSL Behring; she reports consultancy fees from Foresee, Merck, Citius Pharmaceuticals, Quark, and Boehringer Ingelheim, outside of the submitted work.
Figures


References
-
- Ashbaugh DG, Bigelow DB, Petty TL, Levine BE. Acute respiratory distress in adults. Lancet. 1967;2:319–323.
-
- Severinghaus JW. Takuo Aoyagi: discovery of pulse oximetry. Anesth Analg. 2007;105(suppl):S1–S4. - PubMed
-
- Murray JF, Matthay MA, Luce JM, Flick MR. An expanded definition of the adult respiratory distress syndrome. Am Rev Respir Dis. 1988;138:720–723. - PubMed
-
- Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: the Berlin definition. JAMA. 2012;307:2526–2533. - PubMed
-
- Bernard GR, Artigas A, Brigham KL, et al. Report of the American–European consensus conference on ARDS: definitions, mechanisms, relevant outcomes and clinical trial coordination. Intensive Care Med. 1994;20:225–232. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

