A shared mucosal gut microbiota signature in primary sclerosing cholangitis before and after liver transplantation
- PMID: 36056902
- PMCID: PMC9936983
- DOI: 10.1002/hep.32773
A shared mucosal gut microbiota signature in primary sclerosing cholangitis before and after liver transplantation
Abstract
Background and aims: Several characteristic features of the fecal microbiota have been described in primary sclerosing cholangitis (PSC), whereas data on mucosal microbiota are less consistent. We aimed to use a large colonoscopy cohort to investigate key knowledge gaps, including the role of gut microbiota in PSC with inflammatory bowel disease (IBD), the effect of liver transplantation (LT), and whether recurrent PSC (rPSC) may be used to define consistent microbiota features in PSC irrespective of LT.
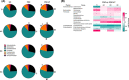
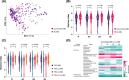
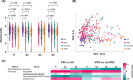
Approach and results: We included 84 PSC and 51 liver transplanted PSC patients (PSC-LT) and 40 healthy controls (HCs) and performed sequencing of the 16S ribosomal RNA gene (V3-V4) from ileocolonic biopsies. Intraindividual microbial diversity was reduced in both PSC and PSC-LT versus HCs. An expansion of Proteobacteria was more pronounced in PSC-LT (up to 19% relative abundance) than in PSC (up to 11%) and HCs (up to 8%; Q FDR < 0.05). When investigating PSC before (PSC vs. HC) and after LT (rPSC vs. no-rPSC), increased variability (dispersion) in the PSC group was found. Five genera were associated with PSC before and after LT. A dysbiosis index calculated from the five genera, and the presence of the potential pathobiont, Klebsiella , were associated with reduced LT-free survival. Concomitant IBD was associated with reduced Akkermansia .
Conclusions: Consistent mucosal microbiota features associated with PSC, PSC-IBD, and disease severity, irrespective of LT status, highlight the usefulness of investigating PSC and rPSC in parallel, and suggest that the impact of gut microbiota on posttransplant liver health should be investigated further.
Copyright © 2023 The Author(s). Published by Wolters Kluwer Health, Inc. on behalf of American Association for the Study of Liver Diseases.
Conflict of interest statement
Tom Hemming Karlsen owns stock in Ultimovacs. He advises for Intercept, Gilead, and Albireo. Kristin Kaasen Jørgensen advises for Celltrion and Norgine. She is on the speakers’ bureau for BMS and Roche.
Figures




Comment in
-
Can a mucosal microbiota signature predict disease severity, survival, and disease recurrence in PSC?Hepatology. 2023 Mar 1;77(3):709-711. doi: 10.1097/HEP.0000000000000021. Epub 2023 Jan 3. Hepatology. 2023. PMID: 36626635 No abstract available.
-
Reply: The mucosal gut signature in primary sclerosing cholangitis before and after liver transplantation. Is the dysbiosis index really predictive for the recurrence of PSC?Hepatology. 2023 Jun 1;77(6):E188-E189. doi: 10.1097/HEP.0000000000000340. Epub 2023 Feb 24. Hepatology. 2023. PMID: 36815351 No abstract available.
-
Letter to the Editor: The mucosal gut signature in primary sclerosing cholangitis before and after liver transplantation. Is the dysbiosis index really predictive for the recurrence of PSC?Hepatology. 2023 Jun 1;77(6):E186-E187. doi: 10.1097/HEP.0000000000000338. Epub 2023 Feb 24. Hepatology. 2023. PMID: 36815357 No abstract available.
-
Letter to the Editor: Mucosal gut microbiota in primary sclerosing cholangitis before and after liver transplantation-Are there other challenges?Hepatology. 2023 Aug 1;78(2):E27-E28. doi: 10.1097/HEP.0000000000000503. Epub 2023 Jun 5. Hepatology. 2023. PMID: 37269114 No abstract available.
-
Mucosa-associated microbiota alterations in primary sclerosing cholangitis (PSC) before and after liver transplantation-who is calling the shots?Hepatobiliary Surg Nutr. 2023 Oct 1;12(5):795-797. doi: 10.21037/hbsn-23-335. Epub 2023 Sep 15. Hepatobiliary Surg Nutr. 2023. PMID: 37886186 Free PMC article. No abstract available.
References
-
- Karlsen TH, Folseraas T, Thorburn D, Vesterhus M. Primary sclerosing cholangitis—a comprehensive review. J Hepatol. 2017;67:1298–323. - PubMed
-
- Boonstra K, Weersma RK, van Erpecum KJ, Rauws EA, Spanier BW, Poen AC, et al. Population‐based epidemiology, malignancy risk, and outcome of primary sclerosing cholangitis. Hepatology. 2013;58:2045–55. - PubMed
-
- Lindström L, Jørgensen KK, Boberg KM, Castedal M, Rasmussen A, Rostved AA, et al. Risk factors and prognosis for recurrent primary sclerosing cholangitis after liver transplantation: a Nordic Multicentre Study. Scand J Gastroenterol. 2018;53:297–304. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

