In depth functional characterization of human induced pluripotent stem cell-derived beta cells in vitro and in vivo
- PMID: 36060810
- PMCID: PMC9428245
- DOI: 10.3389/fcell.2022.967765
In depth functional characterization of human induced pluripotent stem cell-derived beta cells in vitro and in vivo
Abstract
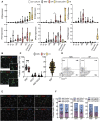
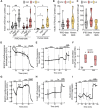
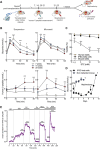
In vitro differentiation of human induced pluripotent stem cells (iPSCs) into beta cells represents an important cell source for diabetes research. Here, we fully characterized iPSC-derived beta cell function in vitro and in vivo in humanized mice. Using a 7-stage protocol, human iPSCs were differentiated into islet-like aggregates with a yield of insulin-positive beta cells comparable to that of human islets. The last three stages of differentiation were conducted with two different 3D culture systems, rotating suspension or static microwells. In the latter, homogeneously small-sized islet-like aggregates were obtained, while in rotating suspension size was heterogeneous and aggregates often clumped. In vitro function was assessed by glucose-stimulated insulin secretion, NAD(P)H and calcium fluctuations. Stage 7 aggregates slightly increased insulin release in response to glucose in vitro. Aggregates were transplanted under the kidney capsule of NOD-SCID mice to allow for further in vivo beta cell maturation. In transplanted mice, grafts showed glucose-responsiveness and maintained normoglycemia after streptozotocin injection. In situ kidney perfusion assays showed modulation of human insulin secretion in response to different secretagogues. In conclusion, iPSCs differentiated with equal efficiency into beta cells in microwells compared to rotating suspension, but the former had a higher experimental success rate. In vitro differentiation generated aggregates lacking fully mature beta cell function. In vivo, beta cells acquired the functional characteristics typical of human islets. With this technology an unlimited supply of islet-like organoids can be generated from human iPSCs that will be instrumental to study beta cell biology and dysfunction in diabetes.
Keywords: aggregate, beta cell; human induced pluripotent stem cell; insulin secretion; islet; microwell.
Copyright © 2022 Fantuzzi, Toivonen, Schiavo, Chae, Tariq, Sawatani, Pachera, Cai, Vinci, Virgilio, Ladriere, Suleiman, Marchetti, Jonas, Gilon, Eizirik, Igoillo-Esteve and Cnop.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

