Additive effect of probiotics (Mutaflor) on 5-aminosalicylic acid therapy in patients with ulcerative colitis
- PMID: 36068716
- PMCID: PMC9449212
- DOI: 10.3904/kjim.2021.458
Additive effect of probiotics (Mutaflor) on 5-aminosalicylic acid therapy in patients with ulcerative colitis
Abstract
Background/aims: In ulcerative colitis (UC) patients, Escherichia coli Nissle 1917 (EcN) is equivalent to mesalazine for preventing disease relapse; however, evidence of the ability of EcN to increase health-related quality of life or induce remission remains scarce. We investigated the efficacy of EcN as an add-on therapy for UC.
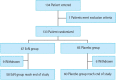
Methods: In this multicentre, double-blind, randomised, placebo-controlled study, a total of 133 UC patients were randomly assigned to receive either EcN or placebo once daily for 8 weeks. Inflammatory bowel disease questionnaire (IBDQ) scores (primary endpoint) and clinical remission and response rates (secondary endpoints) were compared (Clinical trial registration number: NCT04969679).
Results: In total, 118 patients (EcN, 58; placebo, 60) completed the study. The number of patients reaching the primary endpoint did not differ between the EcN and placebo groups (30 [51.7%] vs. 31 [51.7%]; per-protocol analysis, p = 1.0; intention-to-treat analysis, p = 0.86). However, significantly fewer patients in the EcN group exhibited a decreased IBDQ score (1 [1.7%] vs. 8 [13.3%]; per-protocol analysis, p = 0.03; intention- to-treat analysis, p = 0.02). Moreover, a significantly higher number of patients in the EcN group displayed clinical response at 4 weeks (23 [39.7%] vs. 13 [21.7%], p = 0.04) and endoscopic remission at 8 weeks (26 [46.4%] vs. 16 [27.1%], p = 0.03).
Conclusion: Although the number of patients reaching the primary endpoint did not differ between the EcN and placebo groups, EcN was found to be safe and effective in preventing the exacerbation of IBDQ scores and achieving clinical responses and endoscopic remission in patients with mild-to-moderate UC.
Keywords: Colitis, ulcerative; Escherichia coli Nissle 1917; Quality of life.
Conflict of interest statement
This study was supported by a research grant from BL&H Co., Ltd.
Figures
Similar articles
-
Ciprofloxacin and probiotic Escherichia coli Nissle add-on treatment in active ulcerative colitis: a double-blind randomized placebo controlled clinical trial.J Crohns Colitis. 2014 Nov;8(11):1498-505. doi: 10.1016/j.crohns.2014.06.001. Epub 2014 Jun 25. J Crohns Colitis. 2014. PMID: 24972748 Clinical Trial.
-
Clinical trial: probiotic treatment of acute distal ulcerative colitis with rectally administered Escherichia coli Nissle 1917 (EcN).BMC Complement Altern Med. 2010 Apr 15;10:13. doi: 10.1186/1472-6882-10-13. BMC Complement Altern Med. 2010. PMID: 20398311 Free PMC article. Clinical Trial.
-
Therapeutic Potential of Escherichia coli Nissle 1917 in Clinically Remission-attained Ulcerative Colitis Patients: A Hospital-based Cohort Study.Korean J Gastroenterol. 2021 Jan 25;77(1):12-21. doi: 10.4166/kjg.2020.0119. Korean J Gastroenterol. 2021. PMID: 33361702
-
Role and mechanisms of action of Escherichia coli Nissle 1917 in the maintenance of remission in ulcerative colitis patients: An update.World J Gastroenterol. 2016 Jun 28;22(24):5505-11. doi: 10.3748/wjg.v22.i24.5505. World J Gastroenterol. 2016. PMID: 27350728 Free PMC article. Review.
-
Cannabis for the treatment of ulcerative colitis.Cochrane Database Syst Rev. 2018 Nov 8;11(11):CD012954. doi: 10.1002/14651858.CD012954.pub2. Cochrane Database Syst Rev. 2018. PMID: 30406638 Free PMC article.
Cited by
-
Gut Microbiota Modulation in IBD: From the Old Paradigm to Revolutionary Tools.Int J Mol Sci. 2025 Mar 27;26(7):3059. doi: 10.3390/ijms26073059. Int J Mol Sci. 2025. PMID: 40243712 Free PMC article. Review.
-
Bifidobacterium: a probiotic for the prevention and treatment of depression.Front Microbiol. 2023 May 10;14:1174800. doi: 10.3389/fmicb.2023.1174800. eCollection 2023. Front Microbiol. 2023. PMID: 37234527 Free PMC article. Review.
-
Bacterial live therapeutics for human diseases.Mol Syst Biol. 2024 Dec;20(12):1261-1281. doi: 10.1038/s44320-024-00067-0. Epub 2024 Oct 23. Mol Syst Biol. 2024. PMID: 39443745 Free PMC article. Review.
-
Efficacy and safety of probiotics in IBD: An overview of systematic reviews and updated meta-analysis of randomized controlled trials.United European Gastroenterol J. 2024 Sep;12(7):960-981. doi: 10.1002/ueg2.12636. Epub 2024 Aug 6. United European Gastroenterol J. 2024. PMID: 39106167 Free PMC article.
-
New targets for the treatment of ulcerative colitis: Gut microbiota and its metabolites.Comput Struct Biotechnol J. 2025 May 9;27:1850-1863. doi: 10.1016/j.csbj.2025.05.006. eCollection 2025. Comput Struct Biotechnol J. 2025. PMID: 40470314 Free PMC article. Review.
References
-
- Sartor RB. Mechanisms of disease: pathogenesis of Crohn’s disease and ulcerative colitis. Nat Clin Pract Gastroenterol Hepatol. 2006;3:390–407. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical