First-in-human evaluation of [18F]CETO: a novel tracer for adrenocortical tumours
- PMID: 36074157
- PMCID: PMC9816205
- DOI: 10.1007/s00259-022-05957-9
First-in-human evaluation of [18F]CETO: a novel tracer for adrenocortical tumours
Abstract
Purpose: [11C]Metomidate positron emission tomography (PET) is currently used for staging of adrenocortical carcinoma and for lateralization in primary aldosteronism (PA). Due to the short half-life of carbon-11 and a high non-specific liver uptake of [11C]metomidate there is a need for improved adrenal imaging methods. In a previous pre-clinical study para-chloro-2-[18F]fluoroethyletomidate has been proven to be a specific adrenal tracer. The objective is to perform a first evaluation of para-chloro-2-[18F]fluoroethyletomidate positron emission computed tomography ([18F]CETO-PET/CT) in patients with adrenal tumours and healthy volunteers.
Methods: Fifteen patients underwent [18F]CETO-PET/CT. Five healthy volunteers were recruited for test-retest analysis and three out of the five underwent additional [15O]water PET/CT to measure adrenal blood flow. Arterial blood sampling and tracer metabolite analysis was performed. The kinetics of [18F]CETO were assessed and simplified quantitative methods were validated by comparison to outcome measures of tracer kinetic analysis.
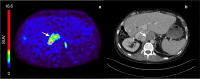
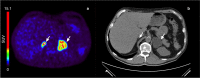
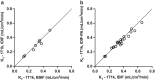
Results: Uptake of [18F]CETO was low in the liver and high in adrenals. Initial metabolization was rapid, followed by a plateau. The kinetics of [18F]CETO in healthy adrenals and all adrenal pathologies, except for adrenocortical carcinoma, were best described by an irreversible single-tissue compartment model. Standardized uptake values (SUV) correlated well with the uptake rate constant K1. Both K1 and SUV were highly correlated to adrenal blood flow in healthy controls. Repeatability coefficients of K1, SUV65-70, and SUV120 were 25, 22, and 17%.
Conclusions: High adrenal uptake combined with a low unspecific liver uptake suggests that 18F]CETO is a suitable tracer for adrenal imaging. Adrenal SUV, based on a whole-body scan at 1 h p.i., correlated well with the net uptake rate Ki.
Trial registration: ClinicalTrials.gov , NCT05361083 Retrospectively registered 29 April 2022. at, https://clinicaltrials.gov/ct2/show/NCT05361083.
Keywords: Adrenal tracer; Positron emission tomography; [18F]CETO.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures








Comment on
-
Para-chloro-2-[18F]fluoroethyl-etomidate: A promising new PET radiotracer for adrenocortical imaging.Int J Med Sci. 2021 Mar 21;18(10):2187-2196. doi: 10.7150/ijms.51206. eCollection 2021. Int J Med Sci. 2021. PMID: 33859526 Free PMC article.
References
-
- Bergström M, Juhlin C, Bonasera TA, Sundin A, Rastad J, Åkerström G, et al. In vitro and in vivo primate evaluation of carbon-11-etomidate and carbon-11- metomidate as potential tracers for PET imaging of the adrenal cortex and its tumors. J Nucl Med. 1998;39:982–989. - PubMed
-
- Burton TJ, Mackenzie IS, Balan K, Koo B, Bird N, Soloviev DV, et al. Evaluation of the sensitivity and specificity of (11)C-metomidate positron emission tomography (PET)-CT for lateralizing aldosterone secretion by Conn's adenomas. J Clin Endocrinol Metab. 2012;97:100–109. doi: 10.1210/jc.2011-1537. - DOI - PubMed
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

