Mainstream genetic testing for women with ovarian cancer provides a solid basis for patients to make a well-informed decision about genetic testing
- PMID: 36076240
- PMCID: PMC9461259
- DOI: 10.1186/s13053-022-00238-w
Mainstream genetic testing for women with ovarian cancer provides a solid basis for patients to make a well-informed decision about genetic testing
Abstract
Background: There is a growing need for genetic testing of women with epithelial ovarian cancer. Mainstream genetic testing provides an alternative care pathway in which non-genetic healthcare professionals offer pre-test counseling themselves. We aimed to explore the impact of mainstream genetic testing on patients' experiences, turnaround times and adherence of non-genetic healthcare professionals to the mainstream genetic testing protocol.
Methods: Patients receiving pre-test counseling at the gynecology departments between April 2018 and April 2020 were eligible to participate in our intervention group. Patients receiving pre-test counseling at the genetics department between January 2017 and April 2020 were eligible to participate in our control group. We evaluated patients' experiences with questionnaires, consisting of questions regarding knowledge, satisfaction and psychosocial outcomes. Patients in the intervention group were sent two questionnaires: one after pre-test counseling and one after receiving their DNA test result. Patients in our control group were sent one questionnaire after receiving their test result. In addition, we collected data regarding turnaround times and adherence of non-genetic healthcare professionals to the mainstream genetic testing protocol.
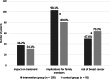
Results: Participation was 79% in our intervention group (105 out of 133 patients) and 60% in our control group (91 out of 152 patients). Knowledge regarding genetics, decisional conflict, depression, anxiety, and distress were comparable in the two groups. In the intervention group, the risk of breast cancer in patients carrying a pathogenic germline variant was discussed less often (49% versus 74% in control group, p ≤ 0.05), and the mean score of regret about the decision to have genetic testing was higher than in the control group (mean 12.9 in the intervention group versus 9.7 in the control group, p ≤ 0.05), although below the clinically relevant threshold of 25. A consent form for the DNA test and a checklist to assess family history were present for ≥ 95% of patients in the intervention group.
Conclusion: Mainstream genetic testing is an acceptable approach to meet the increase in genetic testing among women with epithelial ovarian cancer.
Keywords: Epithelial ovarian cancer; Genetic counseling; Knowledge; Mainstream genetic testing; Patients’ perspectives; Psychosocial outcomes; Satisfaction; Turnaround times.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

References
-
- CRGO . Guideline hereditary and familial ovarian cancer. 2015.
-
- Network NCC . NCCN Clinical Practice Guideline in Oncology: Genetic/Familial High-Risk Assessment: Breast, Ovarian, and Pancreatic, Version 2.2021. 2021. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

