Downregulation of miR-99b-5p and Upregulation of Nuclear mTOR Cooperatively Promotes the Tumor Aggressiveness and Drug Resistance in African American Prostate Cancer
- PMID: 36077039
- PMCID: PMC9455949
- DOI: 10.3390/ijms23179643
Downregulation of miR-99b-5p and Upregulation of Nuclear mTOR Cooperatively Promotes the Tumor Aggressiveness and Drug Resistance in African American Prostate Cancer
Abstract
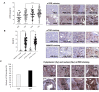
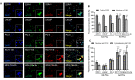
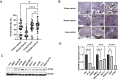
Mammalian target of rapamycin (mTOR) regulates various fundamental cellular events including cell proliferation, protein synthesis, metabolism, apoptosis, and autophagy. Tumor suppressive miR-99b-5p has been implicated in regulating PI3K/AKT/mTOR signaling in a variety of types of cancer. Our previous study suggested the reciprocal miR-99b-5p/MTOR (downregulated/upregulated) pairing as a key microRNA-mRNA regulatory component involved in the prostate cancer (PCa) disparities. In this study, we further validated the expression profiles of mTOR and miR-99b-5p in the PCa, colon, breast, and lung cancer specimens and cell lines. The immunohistochemistry (IHC), immunofluorescence, Western blot, and RT-qPCR assays have confirmed that mTOR is upregulated while miR-99b-5p is downregulated in different patient cohorts and a panel of cancer cell lines. Intriguingly, elevated nuclear mTOR expression was observed in African American PCa and other advanced cancers. Transfection of the miR-99b-5p mimic resulted in a significant reduction in nuclear mTOR and androgen receptor (AR), while a slight/moderate to no decrease in cytoplasmic mTOR and AR in PCa and other cancer cells, suggesting that miR-99b-5p inhibits mTOR and AR expression and their nuclear translocation. Moreover, overexpression of miR-99b-5p targets/inhibits AR-mTOR axis, subsequently initiating cell apoptosis and sensitizing docetaxel-induced cytotoxicity in various cancers. In conclusion, our data suggest that reciprocal miR-99b-5p/nuclear mTOR pairing may be a more precise diagnostic/prognostic biomarker for aggressive PCa, than miR-99b-5p/MTOR pairing or mTOR alone. Targeting the AR-mTOR axis using miR-99b-5p has also been suggested as a novel therapeutic strategy to induce apoptosis and overcome chemoresistance in aggressive PCa.
Keywords: miR-99b-5p; nuclear pmTOR; precision prognostic biomarker; reciprocal miRNA-mRNA pairing.
Conflict of interest statement
These authors declare no conflict of interest.
Figures



Similar articles
-
MicroRNA-99b-5p targets mTOR/AR axis, induces autophagy and inhibits prostate cancer cell proliferation.Tumour Biol. 2022;44(1):107-127. doi: 10.3233/TUB-211568. Tumour Biol. 2022. PMID: 35811549
-
MicroRNA-mRNA Regulatory Network Mediates Activation of mTOR and VEGF Signaling in African American Prostate Cancer.Int J Mol Sci. 2022 Mar 8;23(6):2926. doi: 10.3390/ijms23062926. Int J Mol Sci. 2022. PMID: 35328346 Free PMC article.
-
Tumor suppressive miR-99b-5p as an epigenomic regulator mediating mTOR/AR/SMARCD1 signaling axis in aggressive prostate cancer.Front Oncol. 2023 Nov 7;13:1184186. doi: 10.3389/fonc.2023.1184186. eCollection 2023. Front Oncol. 2023. PMID: 38023145 Free PMC article.
-
Exploring the roles of ncRNAs in prostate cancer via the PI3K/AKT/mTOR signaling pathway.Front Immunol. 2025 Mar 18;16:1525741. doi: 10.3389/fimmu.2025.1525741. eCollection 2025. Front Immunol. 2025. PMID: 40170845 Free PMC article. Review.
-
OncomiR-17-5p: alarm signal in cancer?Oncotarget. 2017 Jul 18;8(41):71206-71222. doi: 10.18632/oncotarget.19331. eCollection 2017 Sep 19. Oncotarget. 2017. PMID: 29050357 Free PMC article. Review.
Cited by
-
Expression pattern, prognostic value and potential microRNA silencing of FZD8 in breast cancer.Oncol Lett. 2023 Sep 21;26(5):477. doi: 10.3892/ol.2023.14065. eCollection 2023 Nov. Oncol Lett. 2023. PMID: 37809047 Free PMC article.
-
Aberrant PI3Kδ splice isoform as a potential biomarker and novel therapeutic target for endocrine cancers.Front Endocrinol (Lausanne). 2023 Aug 21;14:1190479. doi: 10.3389/fendo.2023.1190479. eCollection 2023. Front Endocrinol (Lausanne). 2023. PMID: 37670888 Free PMC article.
-
miRNA regulation of the Akt/mTOR pathway in oral squamous cell carcinoma: a focused review.Discov Oncol. 2025 Jul 17;16(1):1355. doi: 10.1007/s12672-025-03073-2. Discov Oncol. 2025. PMID: 40676313 Free PMC article. Review.
-
PI3K/AKT/mTOR Signaling Pathway in HPV-Driven Head and Neck Carcinogenesis: Therapeutic Implications.Biology (Basel). 2023 Apr 29;12(5):672. doi: 10.3390/biology12050672. Biology (Basel). 2023. PMID: 37237486 Free PMC article. Review.
-
Molecular Insight into Drug Resistance Mechanism Conferred by Aberrant PIK3CD Splice Variant in African American Prostate Cancer.Cancers (Basel). 2023 Feb 20;15(4):1337. doi: 10.3390/cancers15041337. Cancers (Basel). 2023. PMID: 36831678 Free PMC article.
References
-
- Conti D.V., Darst B.V., Moss L.C., Saunders E.J., Sheng X., Chou A., Schumacher F.R., Olama A.A.A., Benlloch S., Dadaev T., et al. Trans-ancestry genome-wide association meta-analysis of prostate cancer identifies new susceptibility loci and informs genetic risk prediction. Nat. Genet. 2021;53:65–75. doi: 10.1038/s41588-020-00748-0. - DOI - PMC - PubMed
-
- Rayford W., Beksac A.T., Alger J., Alshalalfa M., Ahmed M., Khan I., Falagario U.G., Liu Y., Davicioni E., Spratt D.E., et al. Comparative analysis of 1152 African-American and European-American men with prostate cancer identifies distinct genomic and immunological differences. Commun. Biol. 2021;4:670. doi: 10.1038/s42003-021-02140-y. - DOI - PMC - PubMed
-
- Awasthi S., Berglund A., Abraham-Miranda J., Rounbehler R.J., Kensler K., Serna A., Vidal A., You S., Freeman M.R., Davicioni E., et al. Comparative Genomics Reveals Distinct Immune-oncologic Pathways in African American Men with Prostate Cancer. Clin. Cancer. Res. 2021;27:320–329. doi: 10.1158/1078-0432.CCR-20-2925. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

