PTPN2 in the Immunity and Tumor Immunotherapy: A Concise Review
- PMID: 36077422
- PMCID: PMC9456094
- DOI: 10.3390/ijms231710025
PTPN2 in the Immunity and Tumor Immunotherapy: A Concise Review
Abstract
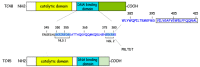
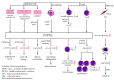
PTPN2 (protein tyrosine phosphatase non-receptor 2), also called TCPTP (T cell protein tyrosine phosphatase), is a member of the PTP family signaling proteins. Phosphotyrosine-based signaling of this non-transmembrane protein is essential for regulating cell growth, development, differentiation, survival, and migration. In particular, PTPN2 received researchers' attention when Manguso et al. identified PTPN2 as a cancer immunotherapy target using in vivo CRISPR library screening. In this review, we attempt to summarize the important functions of PTPN2 in terms of its structural and functional properties, inflammatory reactions, immunomodulatory properties, and tumor immunity. PTPN2 exerts synergistic anti-inflammatory effects in various inflammatory cells and regulates the developmental differentiation of immune cells. The diversity of PTPN2 effects in different types of tumors makes it a potential target for tumor immunotherapy.
Keywords: PTPN2; inflammation; tumor immunotherapy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
PTPN2: Advances and perspectives in cancer treatment potential and inhibitor research.Int J Biol Macromol. 2025 Jun;316(Pt 1):144740. doi: 10.1016/j.ijbiomac.2025.144740. Epub 2025 May 27. Int J Biol Macromol. 2025. PMID: 40441553 Review.
-
Protein tyrosine phosphatase non-receptor type 2 and inflammatory bowel disease.World J Gastroenterol. 2016 Jan 21;22(3):1034-44. doi: 10.3748/wjg.v22.i3.1034. World J Gastroenterol. 2016. PMID: 26811645 Free PMC article. Review.
-
PTPN2 regulates the generation of exhausted CD8+ T cell subpopulations and restrains tumor immunity.Nat Immunol. 2019 Oct;20(10):1335-1347. doi: 10.1038/s41590-019-0480-4. Epub 2019 Sep 16. Nat Immunol. 2019. PMID: 31527834 Free PMC article.
-
Molecular and clinical characterization of PTPN2 expression from RNA-seq data of 996 brain gliomas.J Neuroinflammation. 2018 May 15;15(1):145. doi: 10.1186/s12974-018-1187-4. J Neuroinflammation. 2018. PMID: 29764444 Free PMC article.
-
Protein tyrosine phosphatase nonreceptor type 2 controls colorectal cancer development.J Clin Invest. 2021 Jan 4;131(1):e140281. doi: 10.1172/JCI140281. J Clin Invest. 2021. PMID: 33001862 Free PMC article.
Cited by
-
The HIV latency reversing agent HODHBt inhibits the phosphatases PTPN1 and PTPN2.JCI Insight. 2024 Aug 8;9(18):e179680. doi: 10.1172/jci.insight.179680. JCI Insight. 2024. PMID: 39115957 Free PMC article.
-
STAT3 Signaling Pathway in Health and Disease.MedComm (2020). 2025 Mar 30;6(4):e70152. doi: 10.1002/mco2.70152. eCollection 2025 Apr. MedComm (2020). 2025. PMID: 40166646 Free PMC article. Review.
-
Protein tyrosine phosphatase nonreceptor 2: A New biomarker for digestive tract cancers.World J Gastrointest Oncol. 2025 Feb 15;17(2):100546. doi: 10.4251/wjgo.v17.i2.100546. World J Gastrointest Oncol. 2025. PMID: 39958541 Free PMC article.
-
PTPN2 and Leukopenia in Individuals With Normal TPMT and NUDT15 Metabolizer Status Taking Azathioprine.Clin Transl Sci. 2025 Jun;18(6):e70220. doi: 10.1111/cts.70220. Clin Transl Sci. 2025. PMID: 40442974 Free PMC article.
-
Tumor microenvironment responsive nano-herb and CRISPR delivery system for synergistic chemotherapy and immunotherapy.J Nanobiotechnology. 2024 Jun 19;22(1):346. doi: 10.1186/s12951-024-02571-9. J Nanobiotechnology. 2024. PMID: 38898493 Free PMC article.
References
-
- Penafuerte C., Perez-Quintero L.A., Vinette V., Hatzihristidis T., Tremblay M.L. Mining the Chomplex Family of Protein Tyrosine Phosphatases for Checkpoint Regulators in Immunity. Curr. Top. Microbiol. Immunol. 2017;410:191–214. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

